How to Choose the Right Blood Transfusion Set for Different Scenarios?
May 01,2026
The blood transfusion set is an indispensable sterile medical device designed exclusively for intravenous blood transfusion therapy, which guarantees the safe, controllable, and contamination-free delivery of whole blood or blood components from storage containers to patients’ venous systems. Its standardized design and correct usage are directly associated with the success rate of transfusion and the reduction of adverse reactions, and it has become a basic yet critical tool in emergency treatment, surgical care, hematology departments, and chronic disease management. Without reliable blood transfusion sets, even high-quality blood products cannot be safely infused into patients, increasing the risks of hemolysis, infection, air embolism, and other life-threatening complications.
All qualified blood transfusion sets comply with international and national medical device standards, featuring uniform structural configurations, sterile packaging, and single-use characteristics. Clinical staff must master the complete usage process, inspection criteria, and emergency handling methods of blood transfusion sets to ensure the safety of both medical personnel and patients. This article will systematically elaborate on the composition, classification, usage specifications, quality control, safety precautions, and clinical value of blood transfusion sets to provide a full-range practical guide for medical applications.
Basic Composition and Functional Characteristics of Blood Transfusion Sets
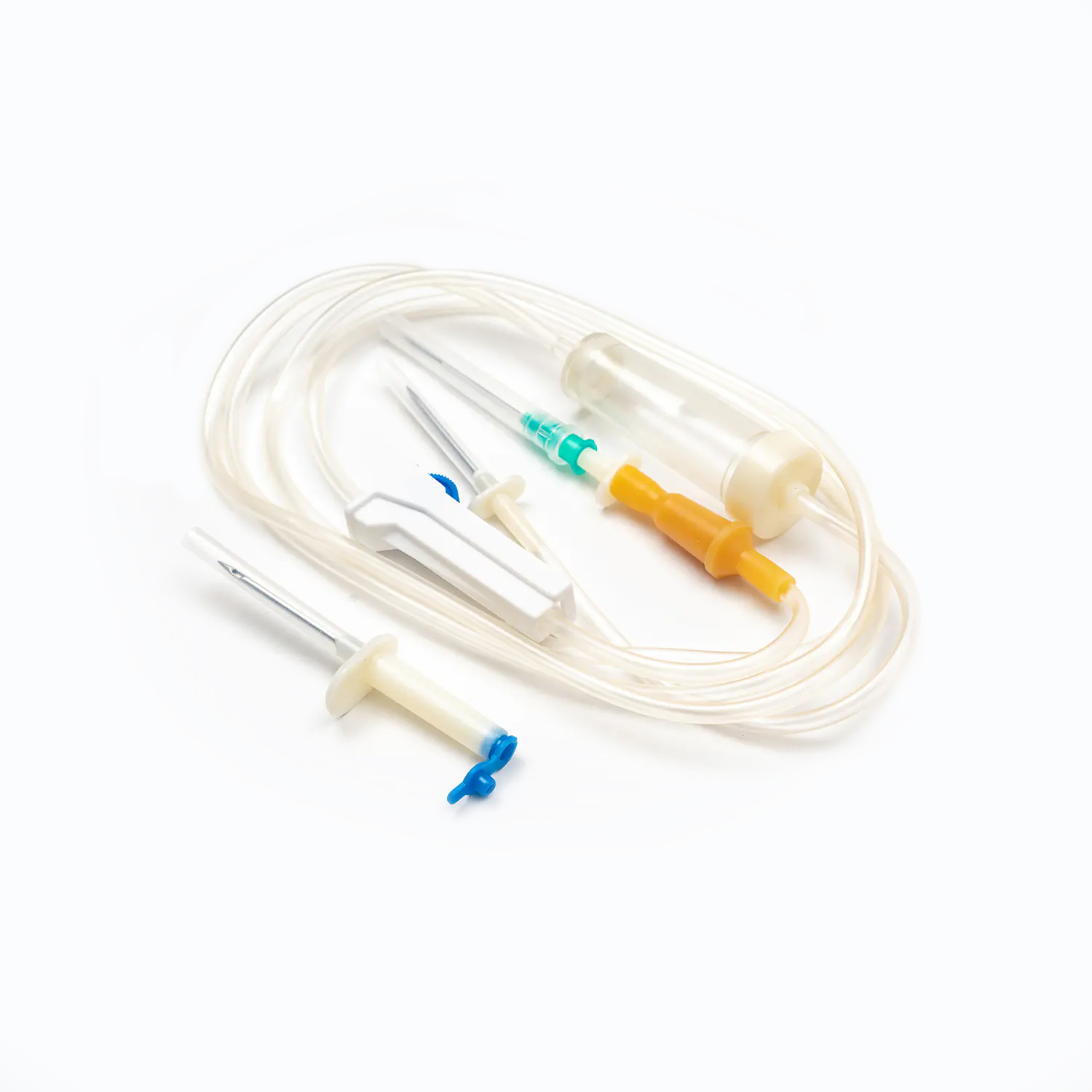
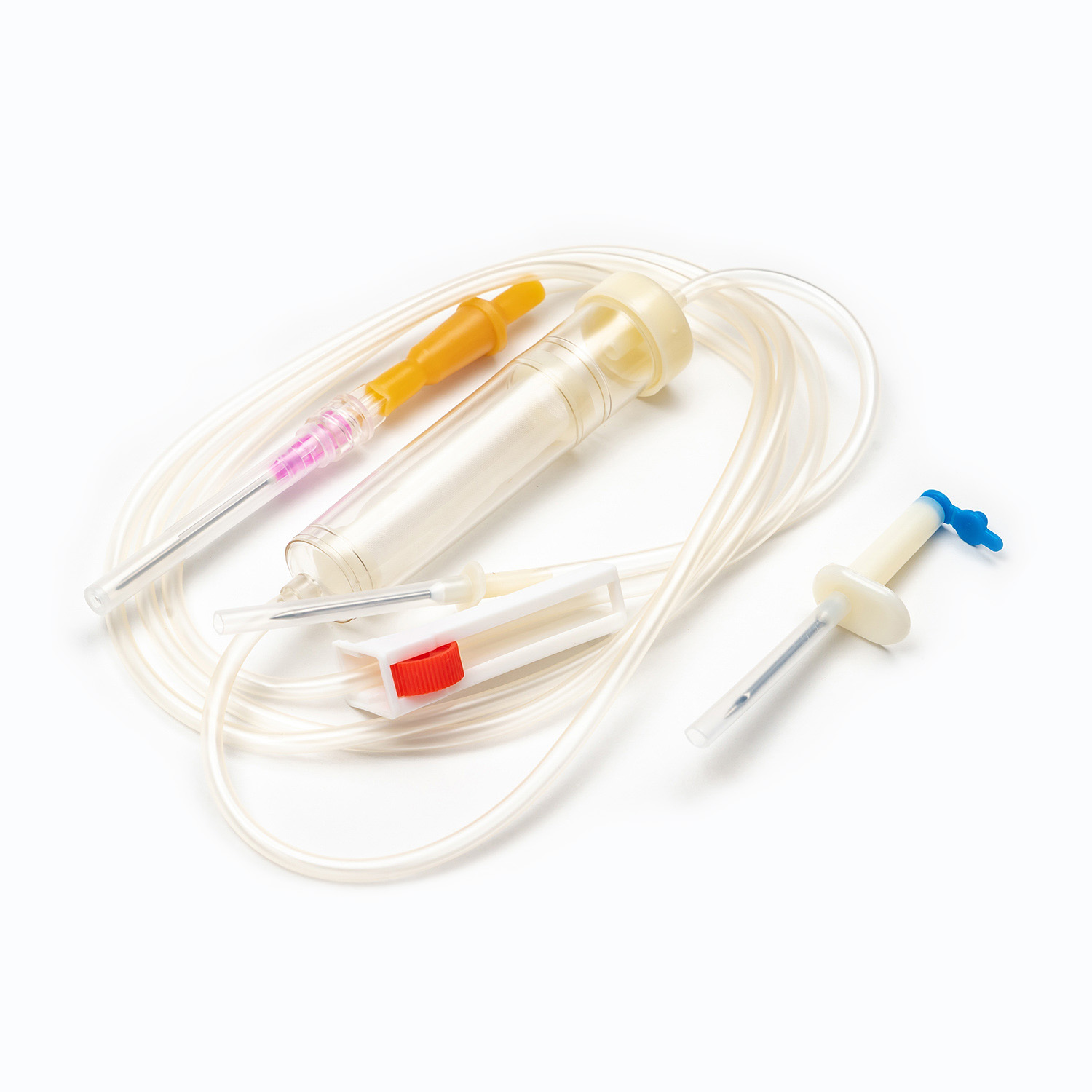
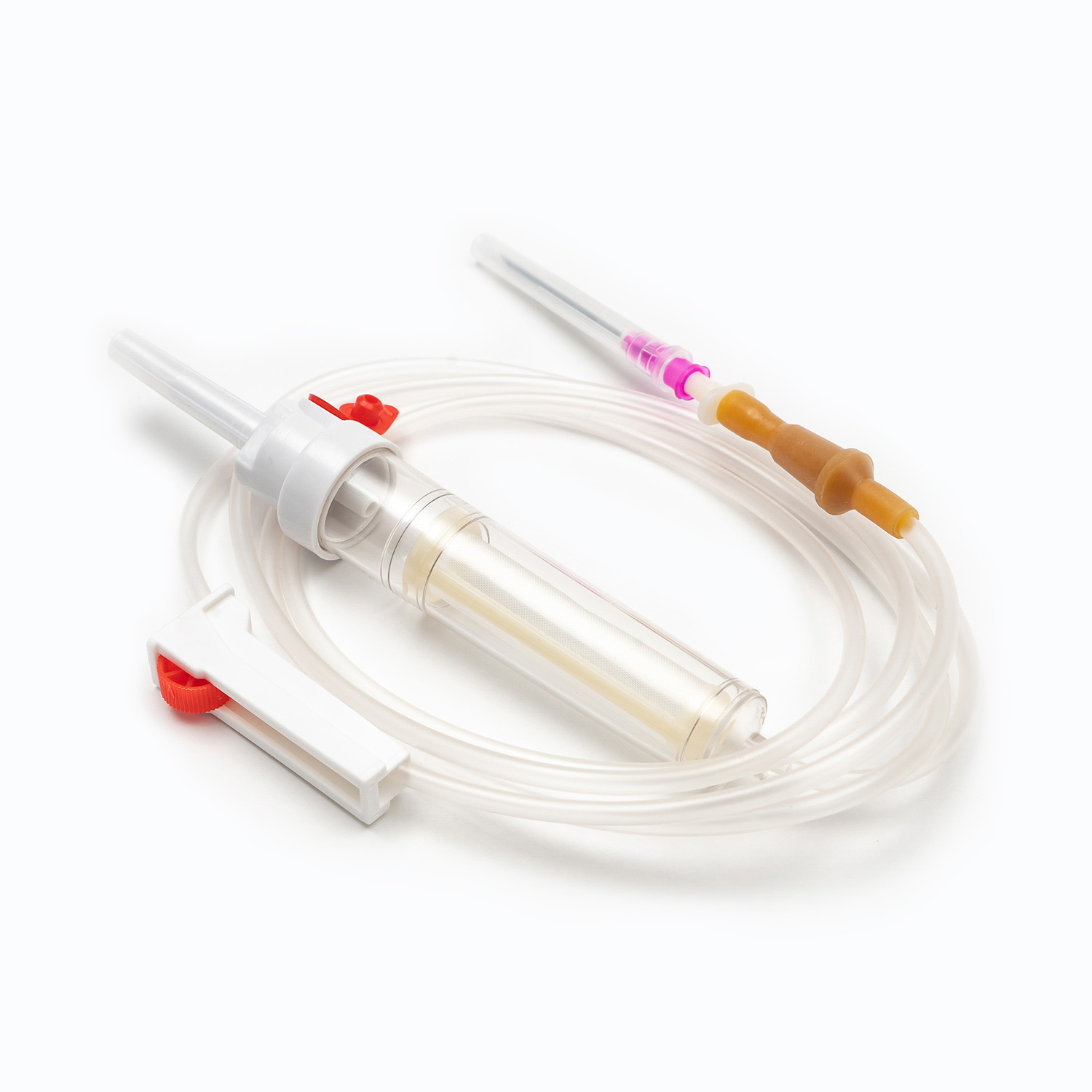
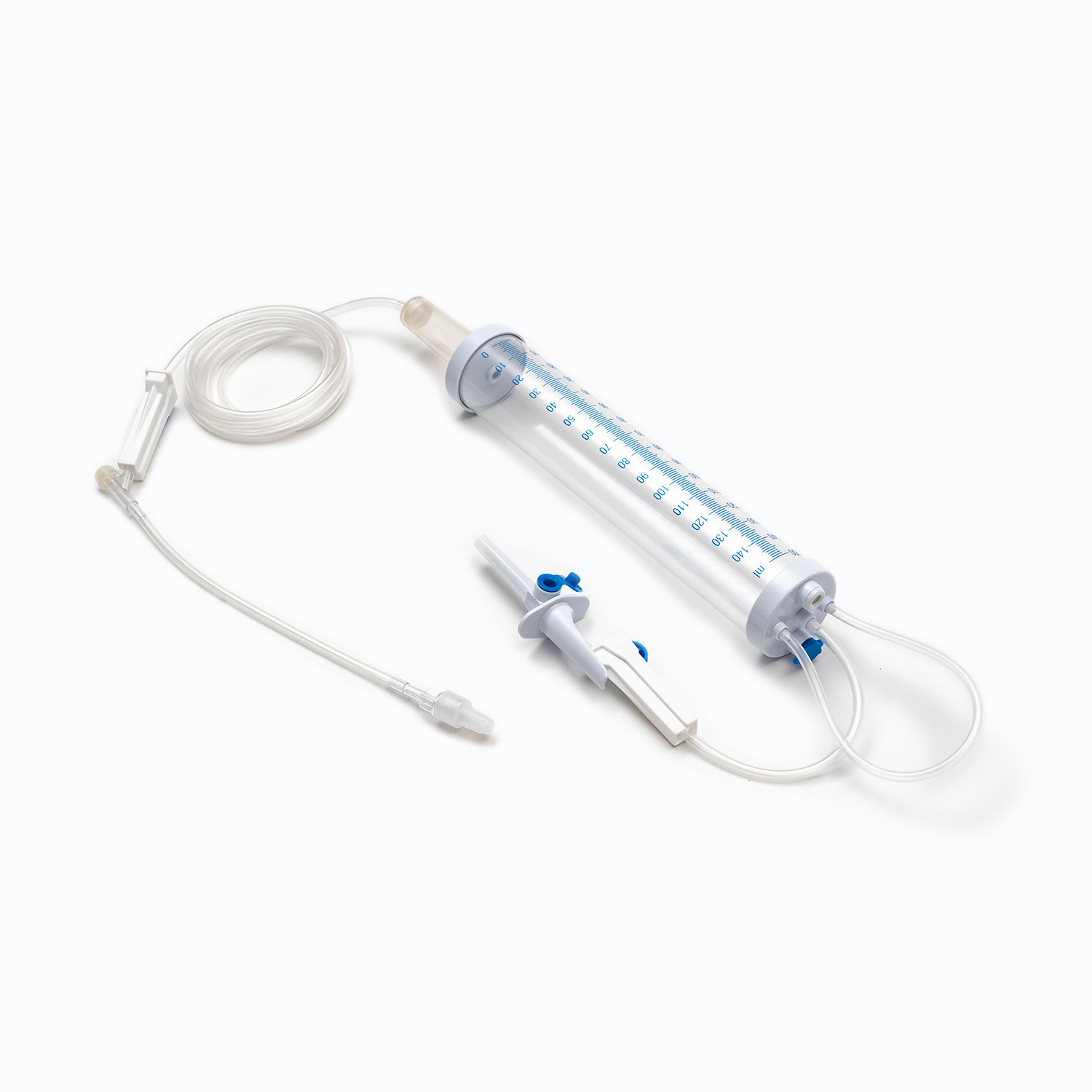
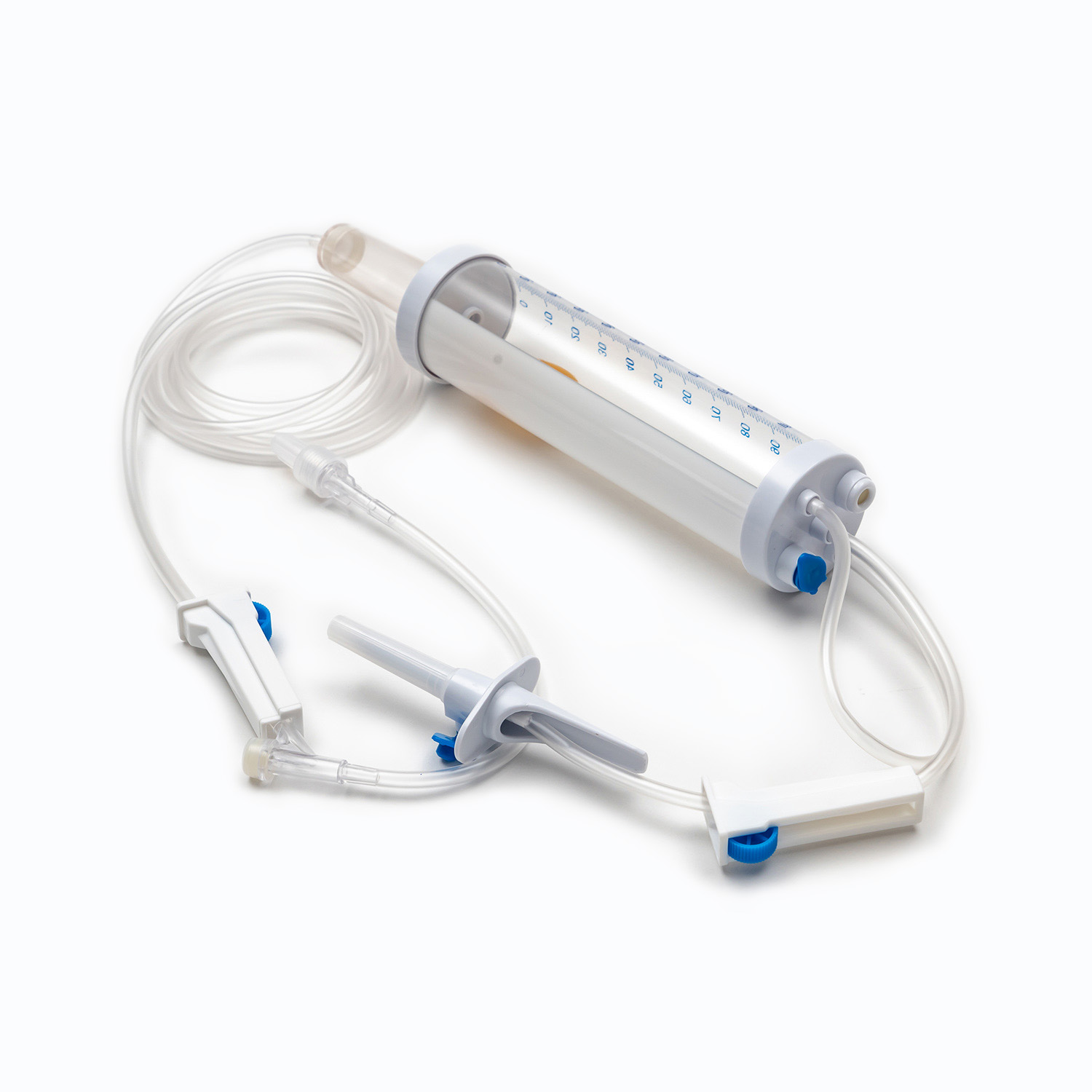
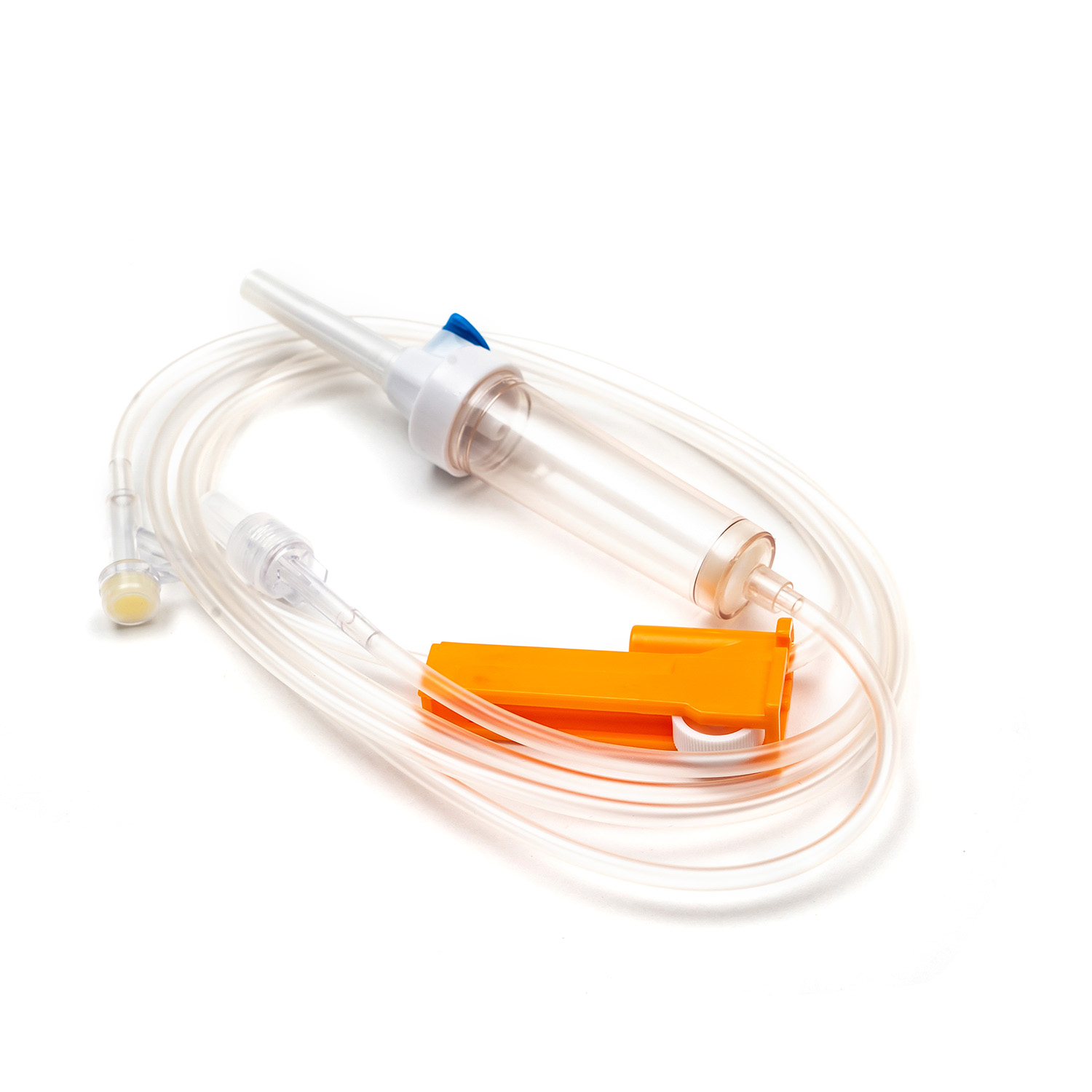
Blood transfusion sets are precision-assembled medical devices composed of multiple independent parts, each undertaking a unique core function. The coordinated operation of all components ensures the stability and safety of the transfusion process. Unlike ordinary infusion sets, blood transfusion sets are specially optimized for blood product characteristics, with enhanced filtration and flow control performance to adapt to the viscosity and cellular components of blood.
Key Component Structure
A standard blood transfusion set consists of the following core components, and each part has strict design requirements:
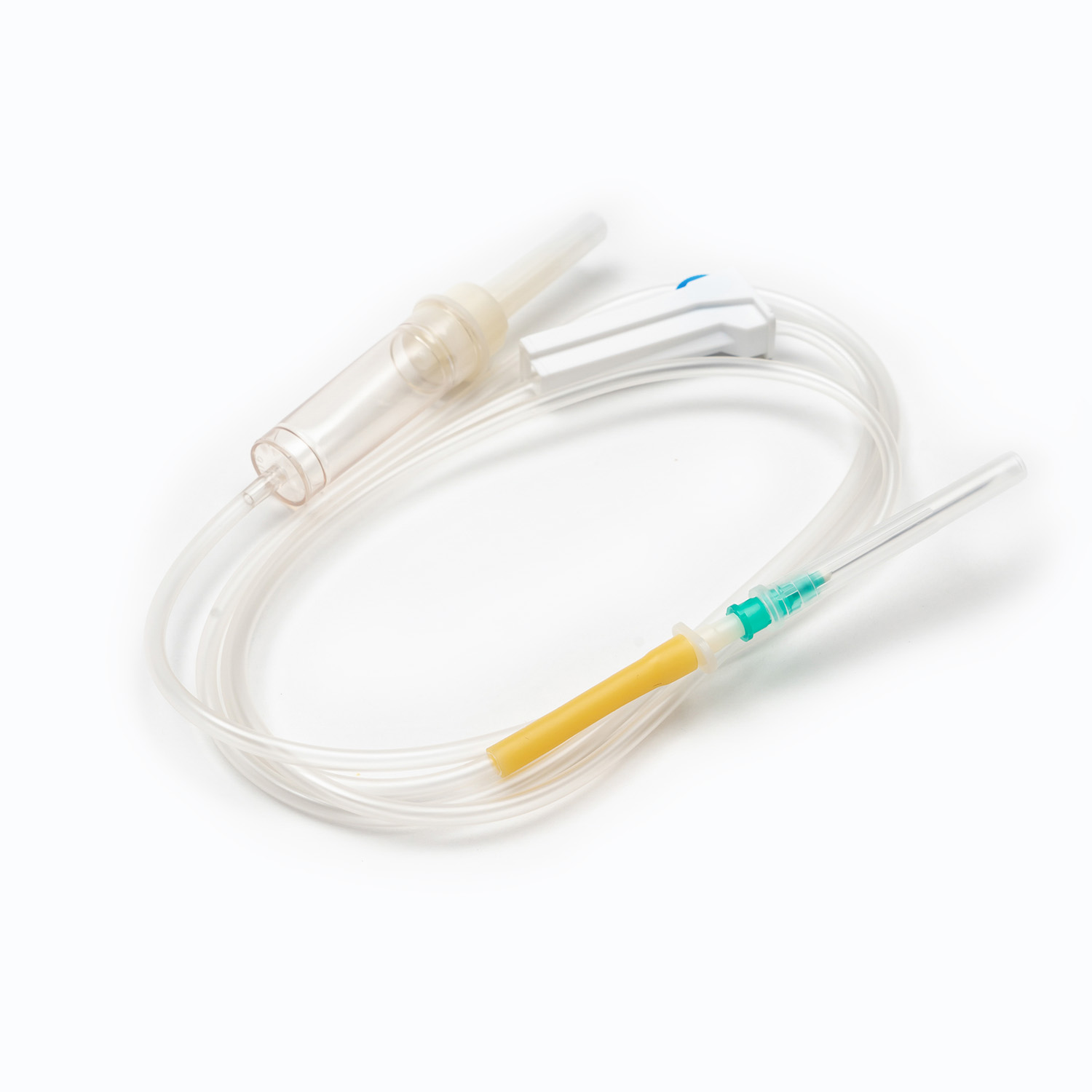
- Spike: The part used to pierce the blood collection bag, made of hard and sharp material, ensuring a tight seal after insertion to prevent blood leakage and air entry.
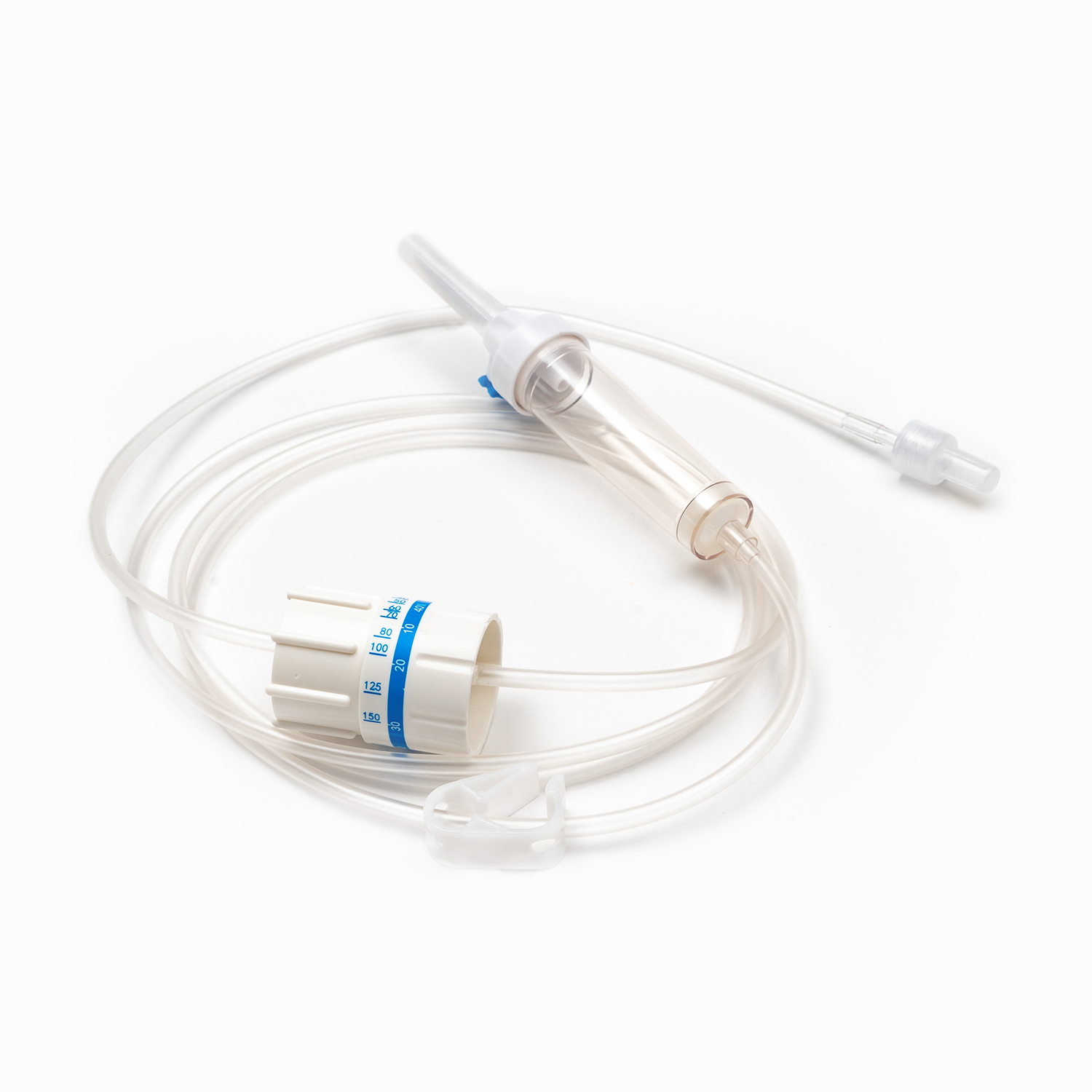
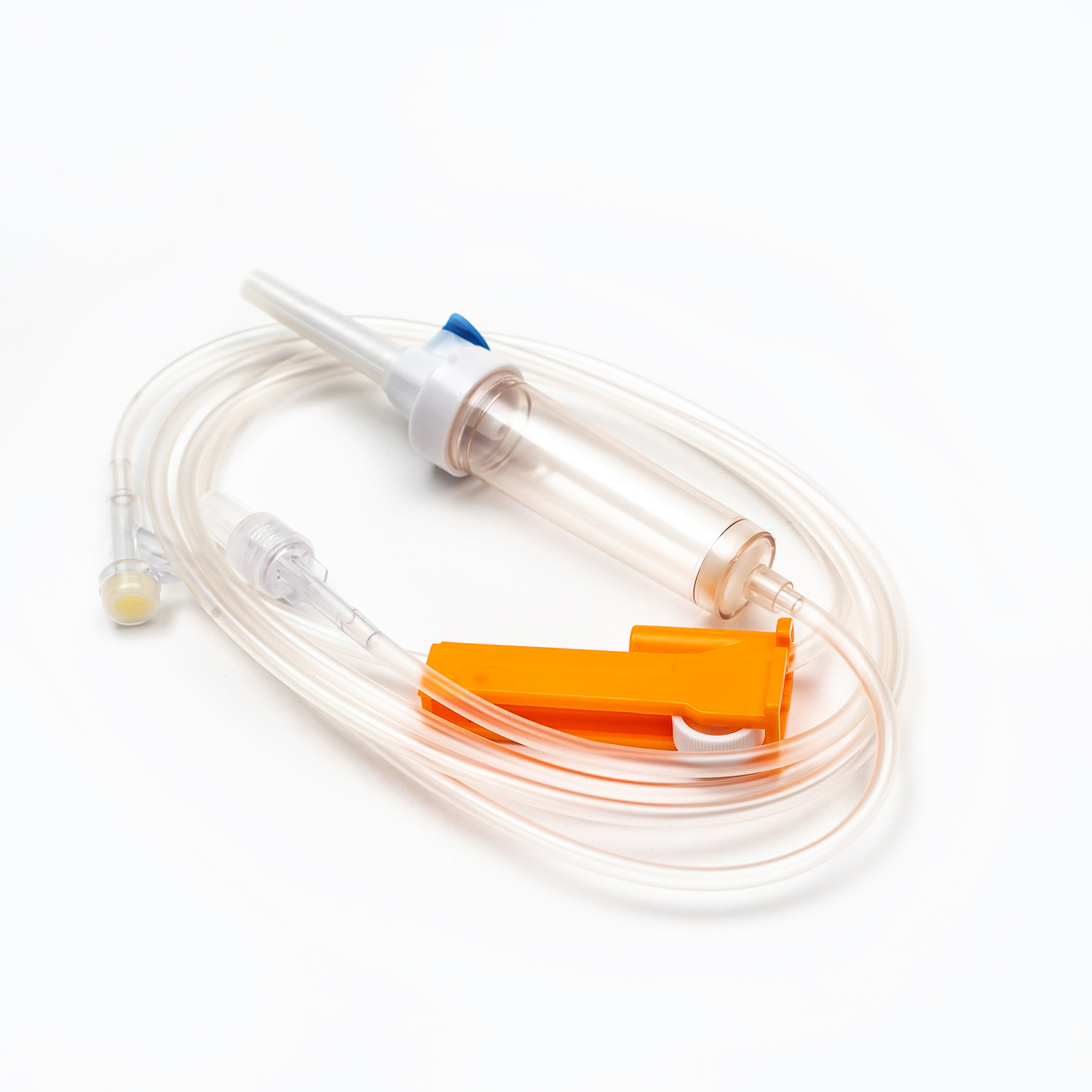
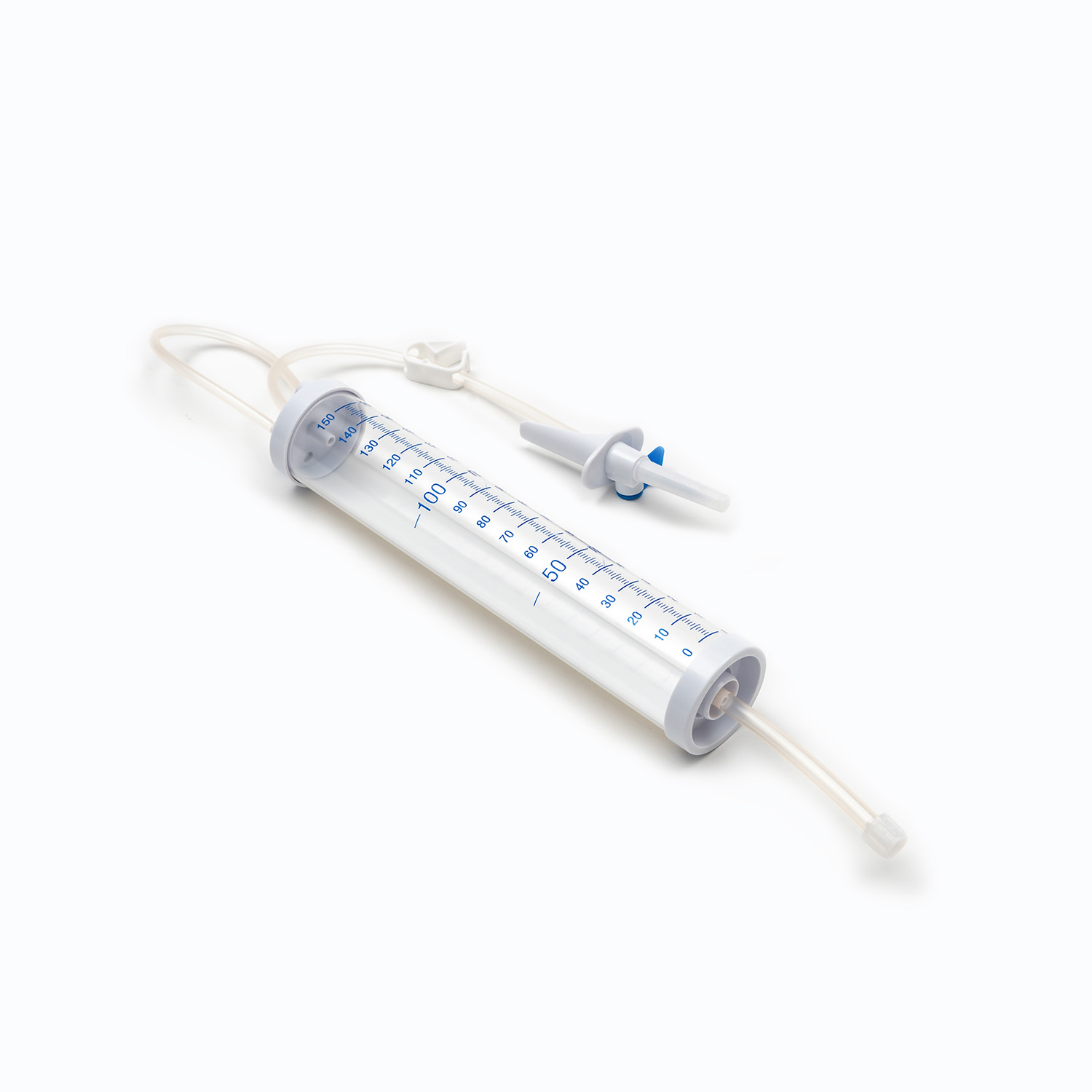
- Drip chamber: A transparent cavity used to observe the transfusion speed, with a quantitative drip function, allowing medical staff to adjust the infusion rate accurately.
- Blood filter: The core component of the transfusion set, with a standard filtration aperture, which can effectively filter out microthrombi, cell aggregates, and impurities in blood products, preventing vascular blockage and adverse reactions.
- Flow regulator: A roller or clip-type device that controls the opening of the infusion tube to adjust the transfusion speed flexibly according to the patient’s condition and doctor’s advice.
- Infusion tube: Made of medical-grade flexible material, non-toxic, non-pyrogenic, and compatible with blood components to avoid hemolysis or blood coagulation.
- Luer connector: A standardized interface used to connect with intravenous indwelling needles or venous catheters, ensuring a firm connection and no fluid leakage.
- Protective cap: Covers the end of the connector to maintain sterility before use and prevent bacterial contamination.
Unique Functional Advantages
Compared with ordinary infusion devices, blood transfusion sets have targeted functional advantages. The built-in filter can remove more than 90% of micro-aggregates in stored blood, which is the most significant difference from infusion sets. The tube material has excellent blood compatibility, which does not damage red blood cells, platelets, and other blood components, maintaining the biological activity of blood products to the greatest extent.
In addition, the transparent design of the entire device allows real-time observation of blood flow, color, and the presence of abnormalities, enabling medical staff to detect problems such as tube blockage, blood coagulation, and air bubbles promptly. The adjustable flow rate function meets the transfusion needs of different groups of people, including adults, children, and critically ill patients, with strong clinical adaptability.
Classification and Application Scenarios of Blood Transfusion Sets
Blood transfusion sets are divided into different types according to clinical needs, structural design, and usage scenarios. Medical institutions select appropriate models based on the patient’s age, condition, and type of blood components infused. Correct classification matching can improve transfusion efficiency and reduce operational risks.
Classification by Target Population
This is the most commonly used classification method in clinical practice, which is divided into adult blood transfusion sets and pediatric blood transfusion sets. The pediatric model has a smaller tube diameter, a more precise flow regulator, and a miniaturized drip chamber to control the transfusion speed accurately and avoid excessive infusion volume in children. The flow control accuracy of pediatric blood transfusion sets is more than twice higher than that of adult models, adapting to the small blood volume and weak tolerance of children.
Classification by Filter Performance
According to the filtration efficiency and aperture, blood transfusion sets are divided into standard filtration types and high-efficiency filtration types. Standard filters are suitable for routine whole blood and red blood cell transfusion, while high-efficiency filters are used for platelet transfusion, fresh frozen plasma, and other sensitive blood components, preventing the loss of effective components while filtering impurities.
Classification by Usage Mode
There are single-use manual blood transfusion sets and automatic transfusion instrument matching sets. Manual models are widely used in general wards and emergency rooms, with simple operation and low cost; automatic matching sets are designed for specialized transfusion pumps, with precise speed control and alarm functions, suitable for operating rooms and intensive care units.
Main Clinical Application Scenarios
| Transfusion Set Type | Applicable Department | Suitable Blood Products |
|---|---|---|
| Standard Adult Type | General Ward, Emergency Room | Whole Blood, Red Blood Cells |
| Pediatric Type | Pediatrics, Neonatology | Component Blood, Small Volume Blood |
| High-Efficiency Filtration Type | Hematology, Oncology | Platelets, Plasma |
| Automatic Pump Type | Operating Room, ICU | Large Volume Rapid Transfusion |
Standardized Usage Procedures of Blood Transfusion Sets
Strictly following standardized operating procedures is the core guarantee for the safe use of blood transfusion sets. Non-standard operations are one of the main causes of transfusion adverse events. All clinical medical staff must complete professional training and assessment before operating blood transfusion sets independently.
Pre-Use Inspection and Preparation
Before use, first check the outer packaging of the blood transfusion set: confirm that the packaging is intact, free of damage and moisture, and check the production date and expiration date. It is strictly prohibited to use expired or damaged blood transfusion sets. Select the appropriate model according to the patient's age and transfusion needs, and prepare matching venous access tools and blood products.
After confirming the validity, open the sterile packaging in a clean environment, take out the blood transfusion set, and avoid touching the spike, filter, and connector parts with hands to prevent contamination. Close the flow regulator in advance to prepare for piercing the blood bag.
Formal Transfusion Operation Steps
- Insert the spike vertically into the blood collection bag port, ensure a tight connection, and hang the blood bag at an appropriate height above the patient's body.
- Squeeze the drip chamber lightly to make blood enter the chamber to half of its volume, then open the flow regulator to exhaust air from the infusion tube until no air bubbles remain in the tube.
- Connect the Luer connector of the transfusion set to the patient's venous indwelling needle, fix it firmly, and adjust the flow regulator to set the initial transfusion speed.
- Observe the patient's vital signs and transfusion status continuously during the transfusion process, and adjust the speed or handle abnormalities promptly.
- After the transfusion is completed, close the flow regulator, pull out the venous access, and dispose of the used blood transfusion set according to medical waste standards.
Key Points of Speed Adjustment
The transfusion speed is determined by the patient's age, cardiac function, and condition. For adult patients with normal cardiopulmonary function, the conventional speed is maintained at a stable range; for elderly patients, children, and patients with heart disease, the speed must be slowed down significantly. Rapid transfusion in a short period of time may increase the risk of heart failure and pulmonary edema in vulnerable groups. For emergency patients with massive hemorrhage, rapid transfusion can be carried out under monitoring to save lives.
Quality Control and Sterility Requirements of Blood Transfusion Sets
As a Class of medical devices in direct contact with blood and human veins, blood transfusion sets have extremely strict quality control and sterility standards, which are supervised by national medical device regulatory agencies. Qualified products must pass a full range of performance and safety tests before being put into clinical use.
Sterility and Pyrogen-Free Standards
All blood transfusion sets must be sterilized by an official method before leaving the factory, with a sterility assurance rate of 100%, and no pyrogens, microorganisms, or harmful residues are allowed. Pyrogens are the main cause of febrile transfusion reactions, so the production process must strictly implement a pyrogen-free control system. The single-use design eliminates the risk of cross-infection caused by repeated use, which is a mandatory requirement for global medical institutions.
Material Safety and Biocompatibility
The production materials of blood transfusion sets are all medical-grade polymer materials, which have passed hemocompatibility tests, including hemolysis test, coagulation test, and cytotoxicity test. The materials do not cause red blood cell rupture, platelet aggregation, or tissue irritation, ensuring that blood components maintain their original activity during infusion.
The tube has good tensile and bending resistance, will not break or block during use, and has stable chemical properties, no precipitation of harmful substances, and no chemical reaction with blood products.
Filtration Performance and Structural Quality Control
The filter of the blood transfusion set has a fixed aperture standard, which can effectively intercept micro-aggregates while ensuring smooth blood flow. The drip chamber, flow regulator, and connector all have uniform dimensional standards to ensure consistent performance of each product. Each batch of products must undergo sampling inspection for flow rate, filtration efficiency, and sealing performance, and unqualified batches are completely destroyed.
Safety Precautions and Adverse Event Handling
Even with high-quality blood transfusion sets and standardized operations, potential risks still exist in the transfusion process. Clinical staff must be familiar with common safety precautions and emergency handling methods to avoid the deterioration of adverse events and protect patients' lives.
Non-Negotiable Safety Precautions
- Single-use only: Do not clean, disinfect, or reuse blood transfusion sets to prevent cross-infection and structural damage.
- No air mixing: Ensure complete exhaust of air bubbles before transfusion, air embolism caused by air entering the vein is a fatal risk, and must be strictly prevented.
- Compatibility check: Do not mix blood with other drugs through the transfusion set to avoid blood coagulation, hemolysis, or chemical reactions.
- Real-time monitoring: Patients must be observed for at least 15 minutes at the beginning of transfusion, and the most severe adverse reactions occur in this stage.
- Storage conditions: Store blood transfusion sets in a dry, cool, and light-proof environment, away from high temperatures and sharp objects to avoid packaging damage.
Handling of Common Abnormal Situations
When blood flow is blocked during transfusion, first check whether the infusion tube is folded or the filter is blocked, and replace the blood transfusion set immediately if necessary. If the patient has fever, rash, dyspnea, and other allergic or febrile reactions, stop the transfusion immediately, keep the venous access unobstructed, and notify the doctor for emergency treatment.
If blood leakage occurs at the connector or spike, close the flow regulator immediately and replace the transfusion set to avoid blood loss and contamination. For all adverse events related to the blood transfusion set, complete records should be kept and reported to the medical device management department in accordance with regulations.
Clinical Value and Development Trend of Blood Transfusion Sets
Blood transfusion is one of the important means of modern medical treatment, and blood transfusion sets, as its carrier device, have irreplaceable clinical value. They are widely used in various medical scenarios and play a vital role in saving lives and treating diseases.
Core Clinical Value
In emergency trauma treatment, blood transfusion sets achieve rapid blood transfusion to supplement blood volume and treat hemorrhagic shock, increasing the survival rate of patients with acute massive hemorrhage by a large margin. In surgical operations, they support intraoperative blood transfusion to maintain the patient's circulatory stability and ensure the smooth progress of the operation. In hematology, oncology, and other chronic disease treatments, they provide long-term component blood transfusion support to improve patients' anemia symptoms and quality of life.
In addition, blood transfusion sets are also used in blood exchange transfusion for newborns, hematopoietic stem cell transplantation, and other special treatments, with a wide range of applications and high clinical dependence. Their popularization and standardized use have greatly reduced the mortality rate of transfusion-related adverse reactions and promoted the development of transfusion medicine.
Future Development Trend
With the development of medical technology, blood transfusion sets are developing in the direction of intelligence, miniaturization, and high safety. New-type blood transfusion sets are equipped with air detection and anti-reflux functions, which can automatically alarm and block the pipeline when air bubbles are detected, further reducing operational risks.
The filter technology is continuously upgraded, with higher filtration efficiency and lower component loss, adapting to more types of blood products. The lightweight and flexible design makes the operation more convenient, especially suitable for field rescue and mobile medical scenarios. In the future, blood transfusion sets will be more integrated with intelligent transfusion equipment to achieve fully automated and precise transfusion management.
At the same time, the production process pays more attention to environmental protection and sustainability, using eco-friendly materials while ensuring safety, reducing the impact of medical waste on the environment. The standardization and internationalization of blood transfusion sets are also deepening, promoting the consistency of transfusion safety standards worldwide and benefiting more patients.



 English
English Français
Français русский
русский Español
Español