Why Are Disposable Infusion Sets Essential in Hospital Treatment?
Apr 22,2026
The disposable infusion set is a single-use, sterile medical device designed for the safe and standardized delivery of intravenous fluids, medications, nutrients, and blood products directly into a patient’s venous system. It is an irreplaceable basic tool in clinical treatment, emergency rescue, chronic disease management, and perioperative care, and its core value lies in preventing cross-infection, ensuring medication delivery accuracy, and improving the safety and efficiency of clinical infusion therapy.
Strict compliance with medical standards, correct selection, standardized operation, and proper disposal are essential to maximize the performance of disposable infusion sets and avoid adverse events such as infection, infusion leakage, and vascular irritation. As a consumable widely used in global medical systems, it meets universal clinical needs while adapting to specialized treatment scenarios, making it one of the most widely applied medical devices in modern healthcare.
Basic Composition and Functional Principles of Disposable Infusion Sets
Main Structural Components and Their Roles
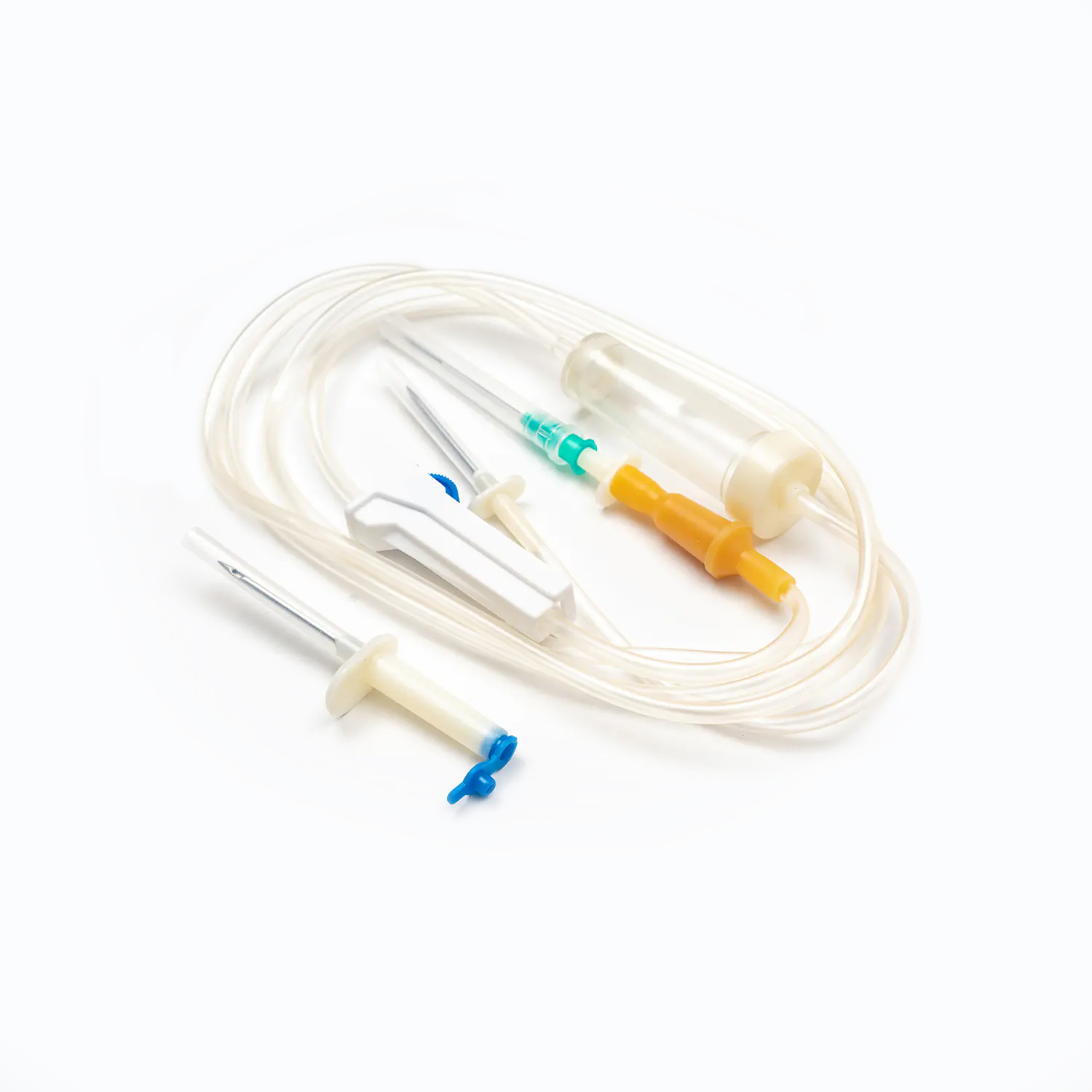
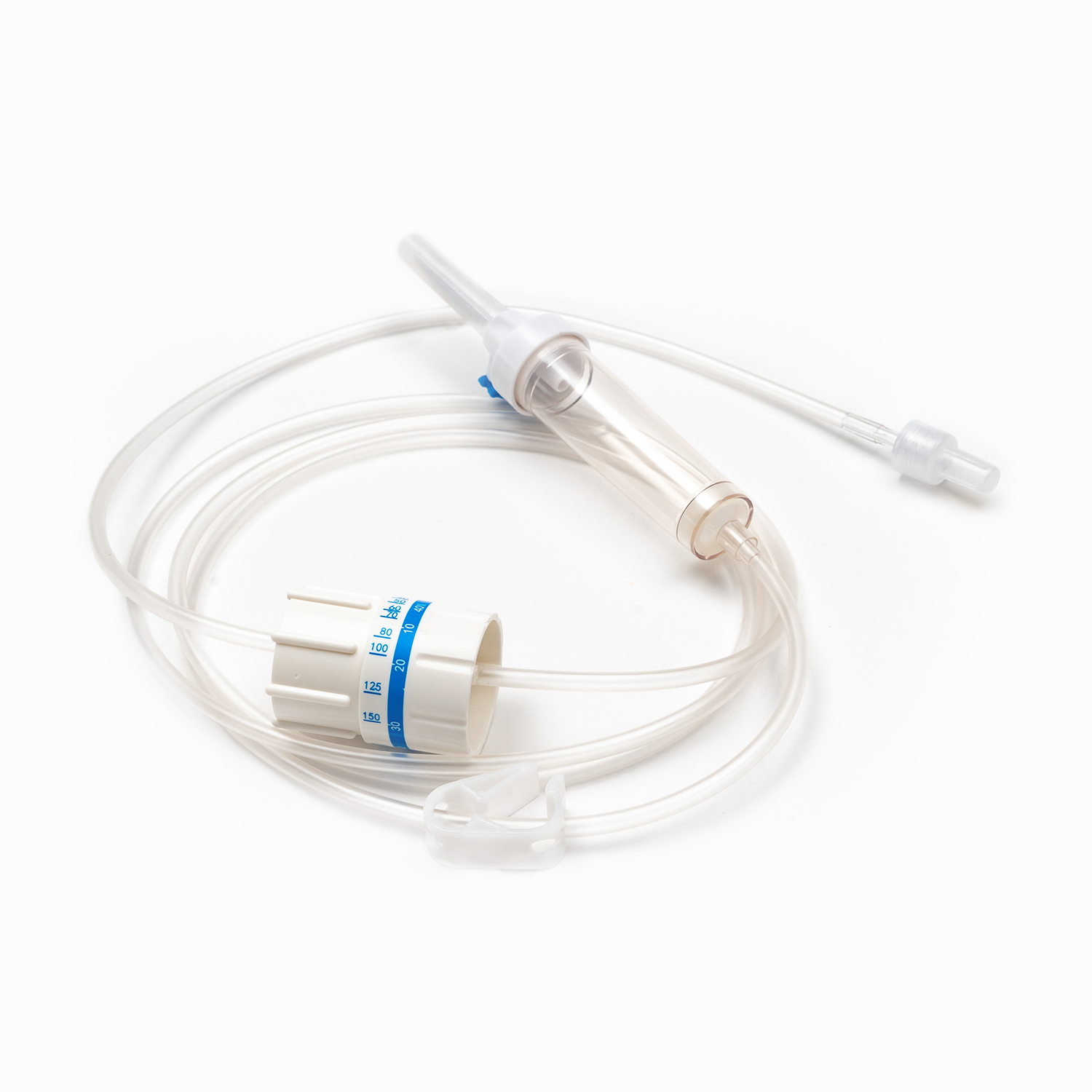
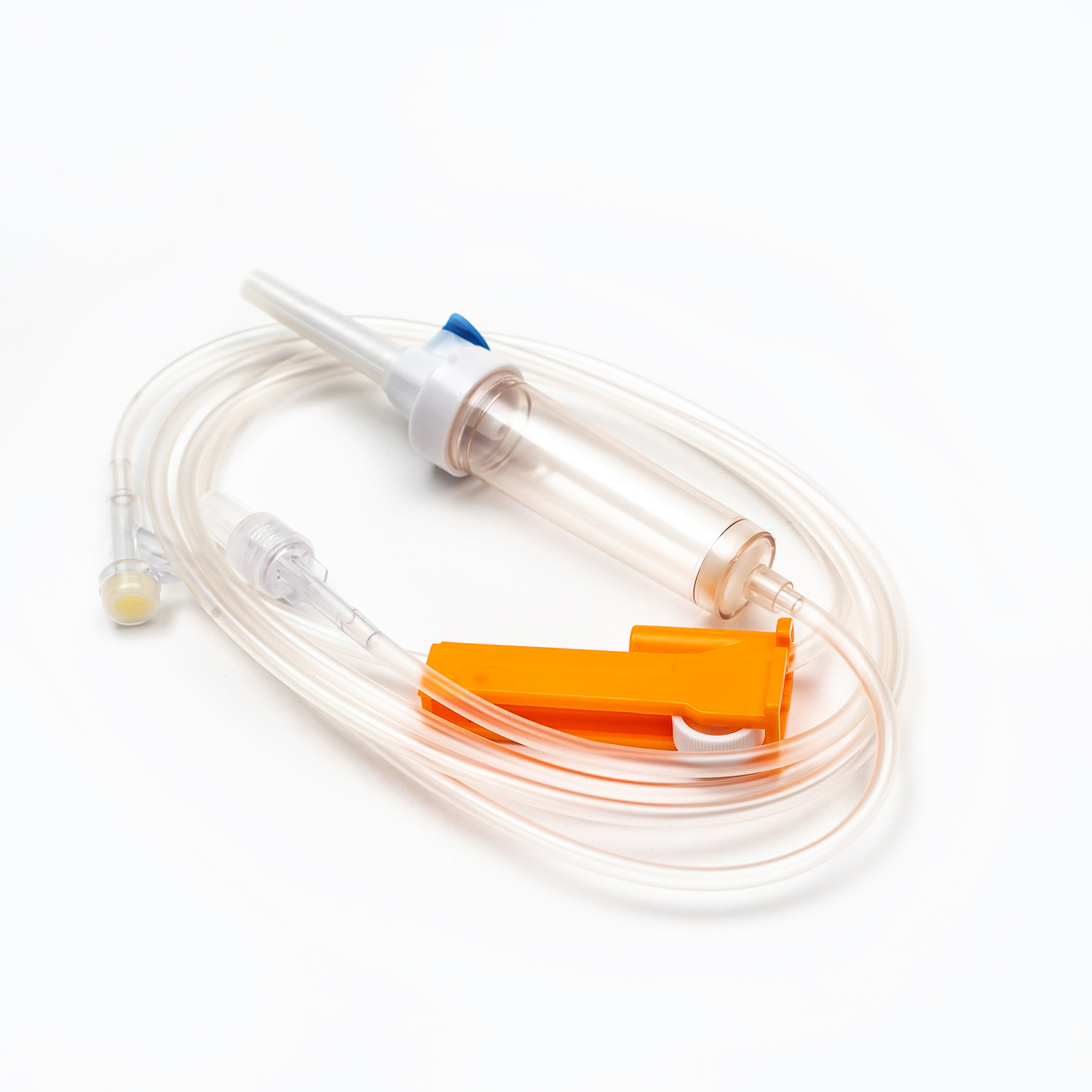
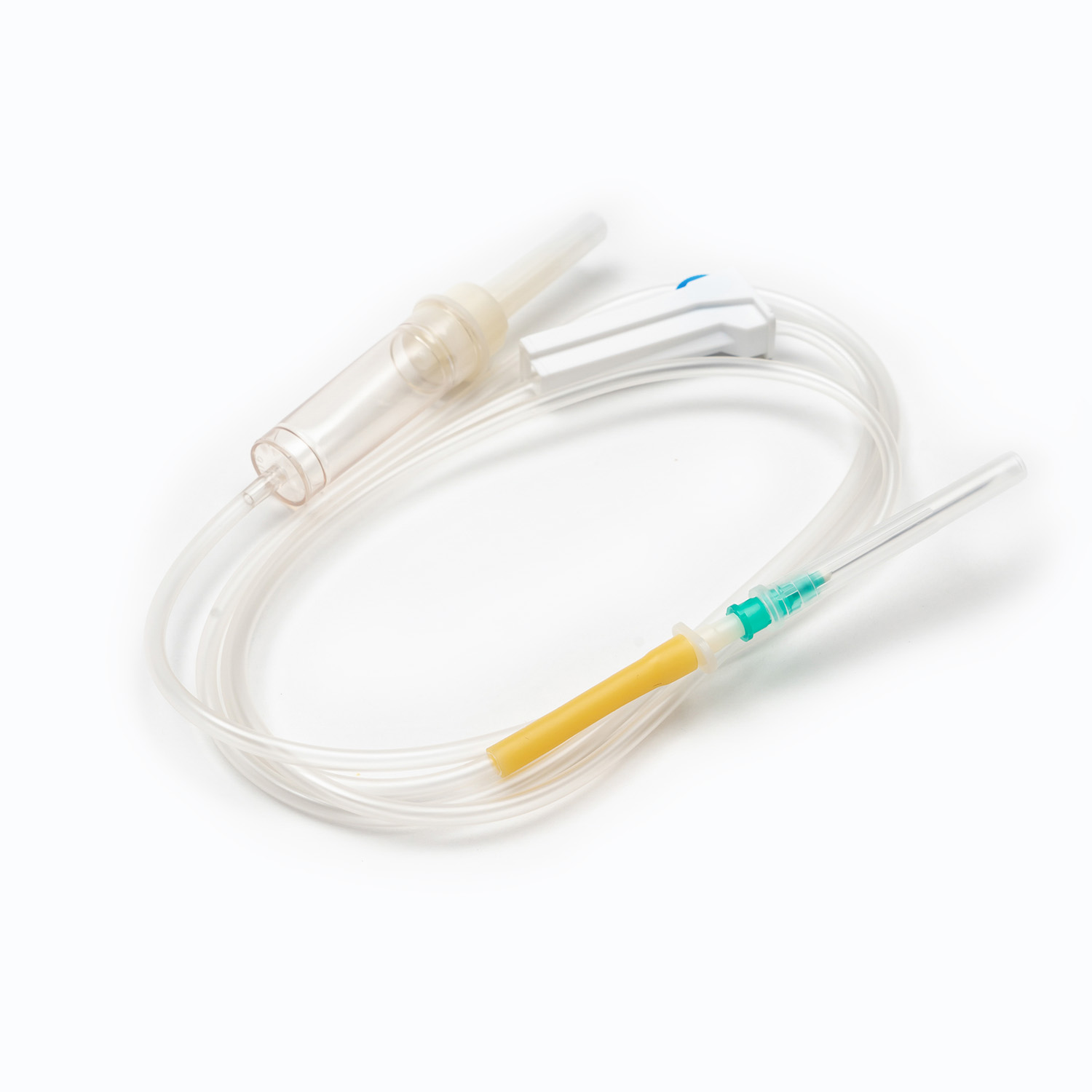
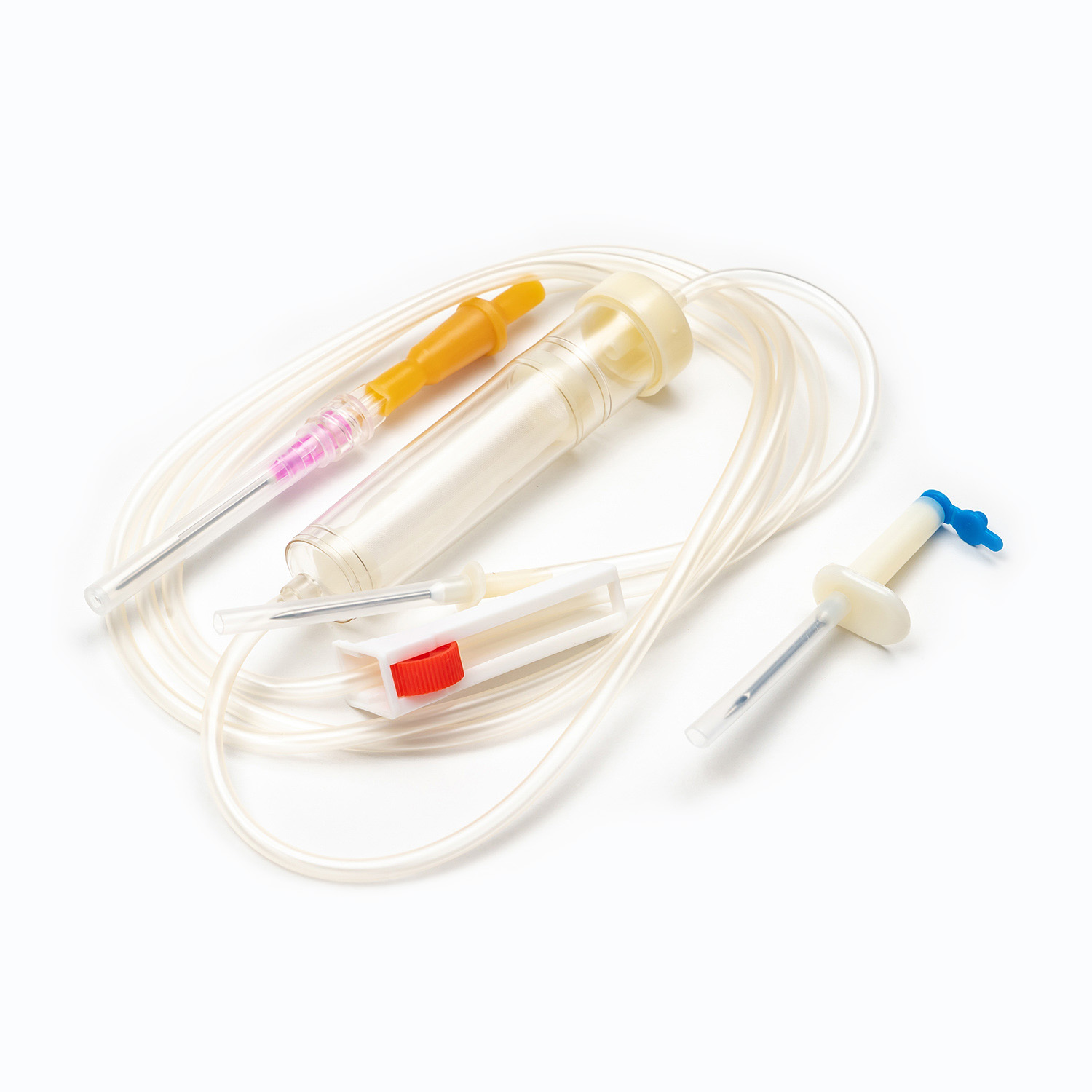
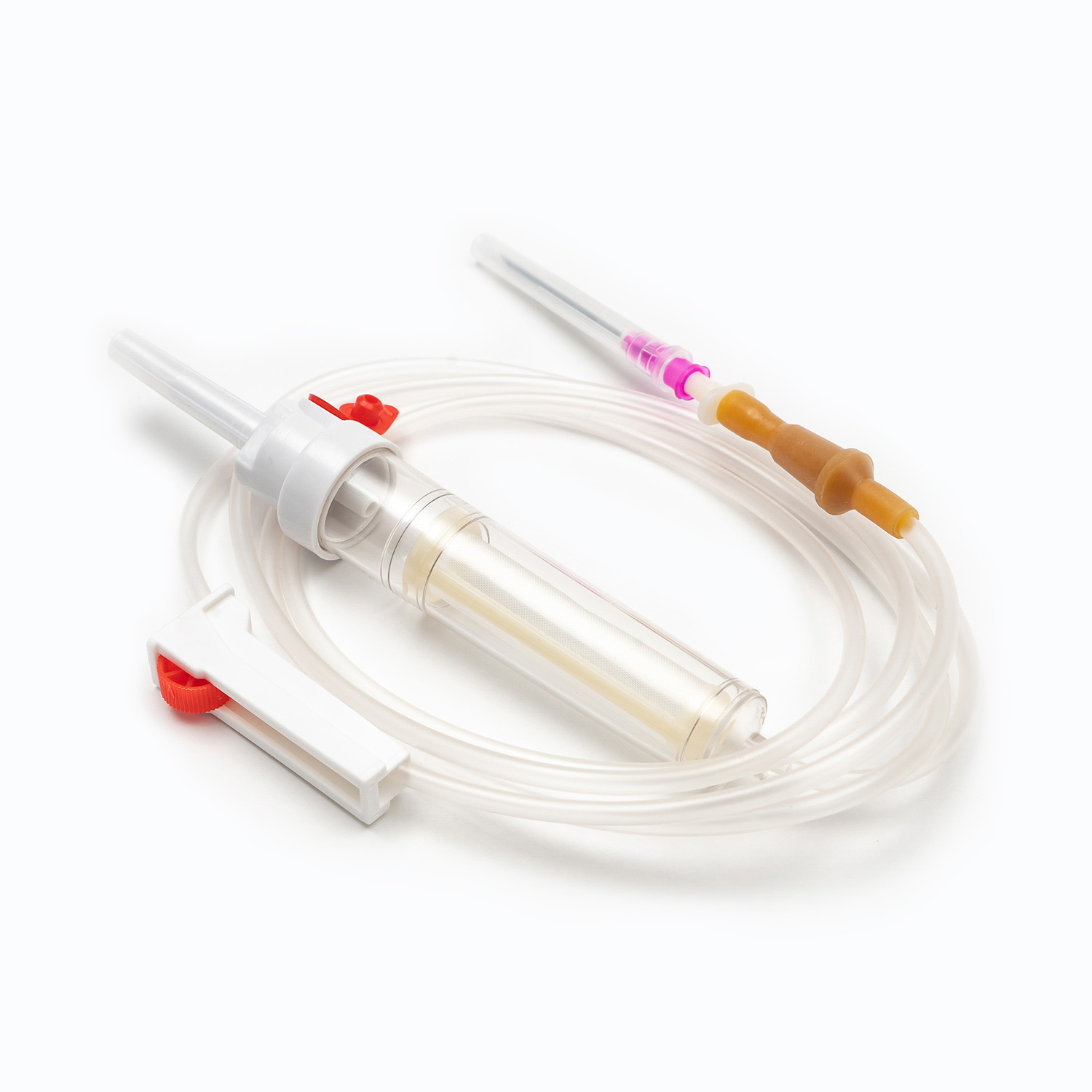
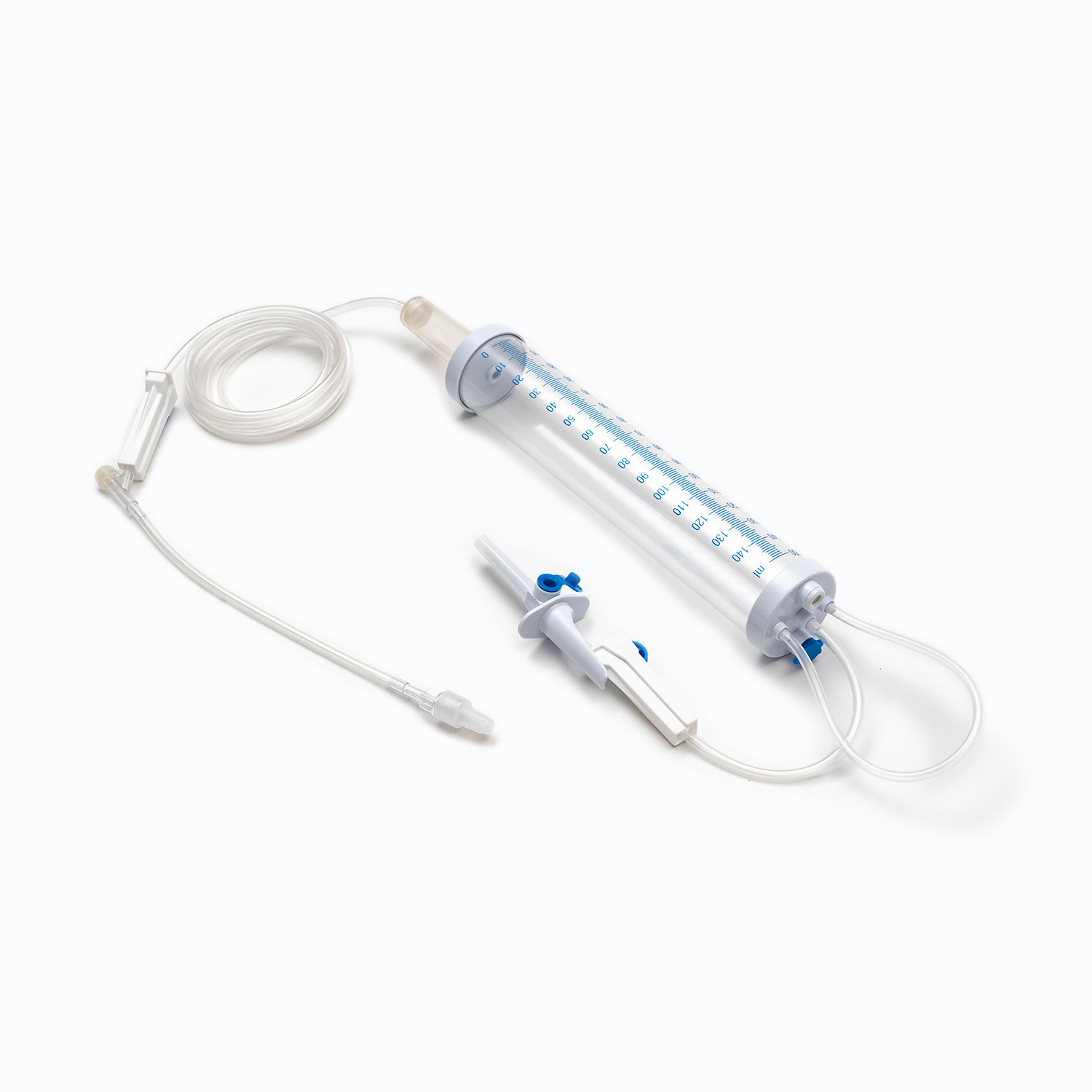
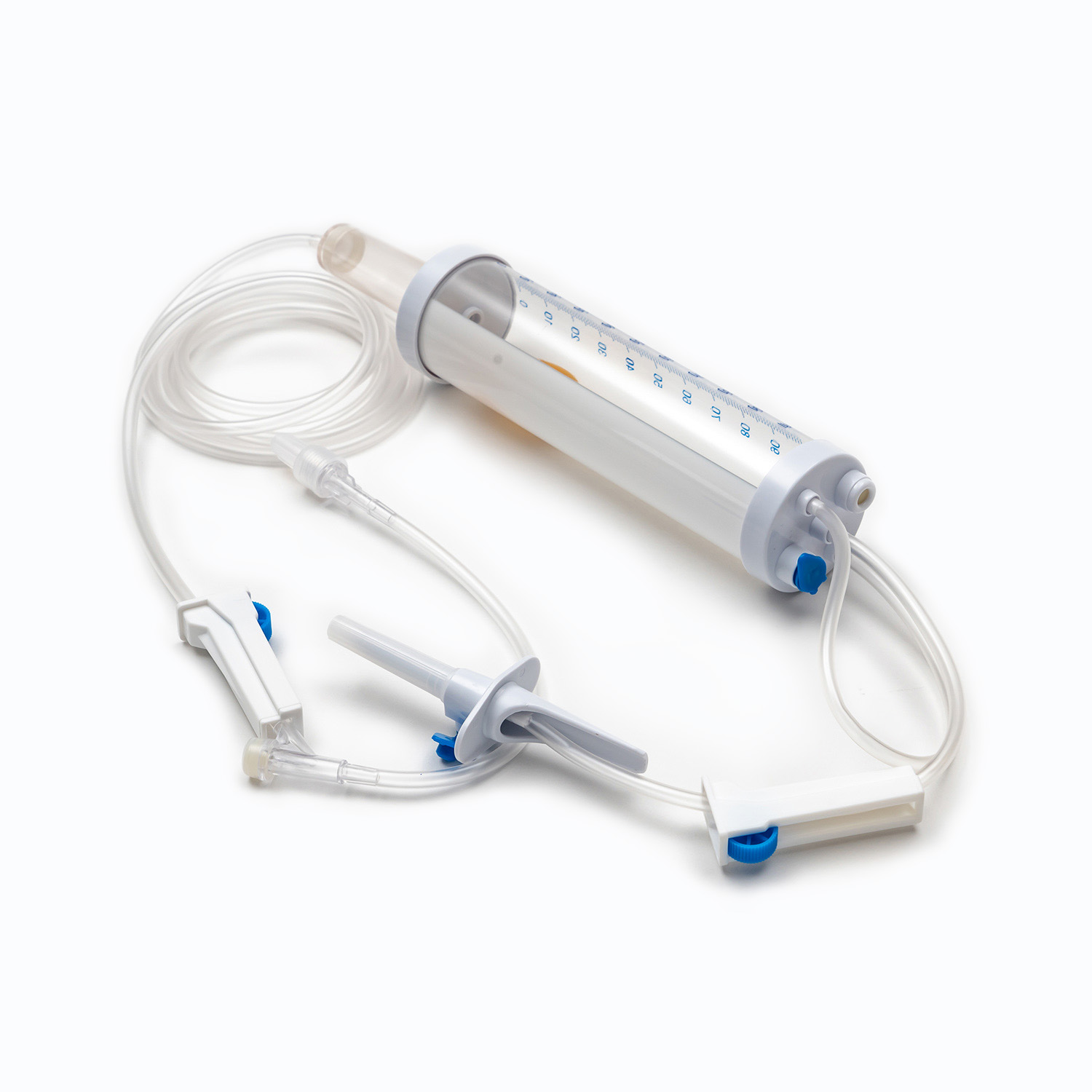
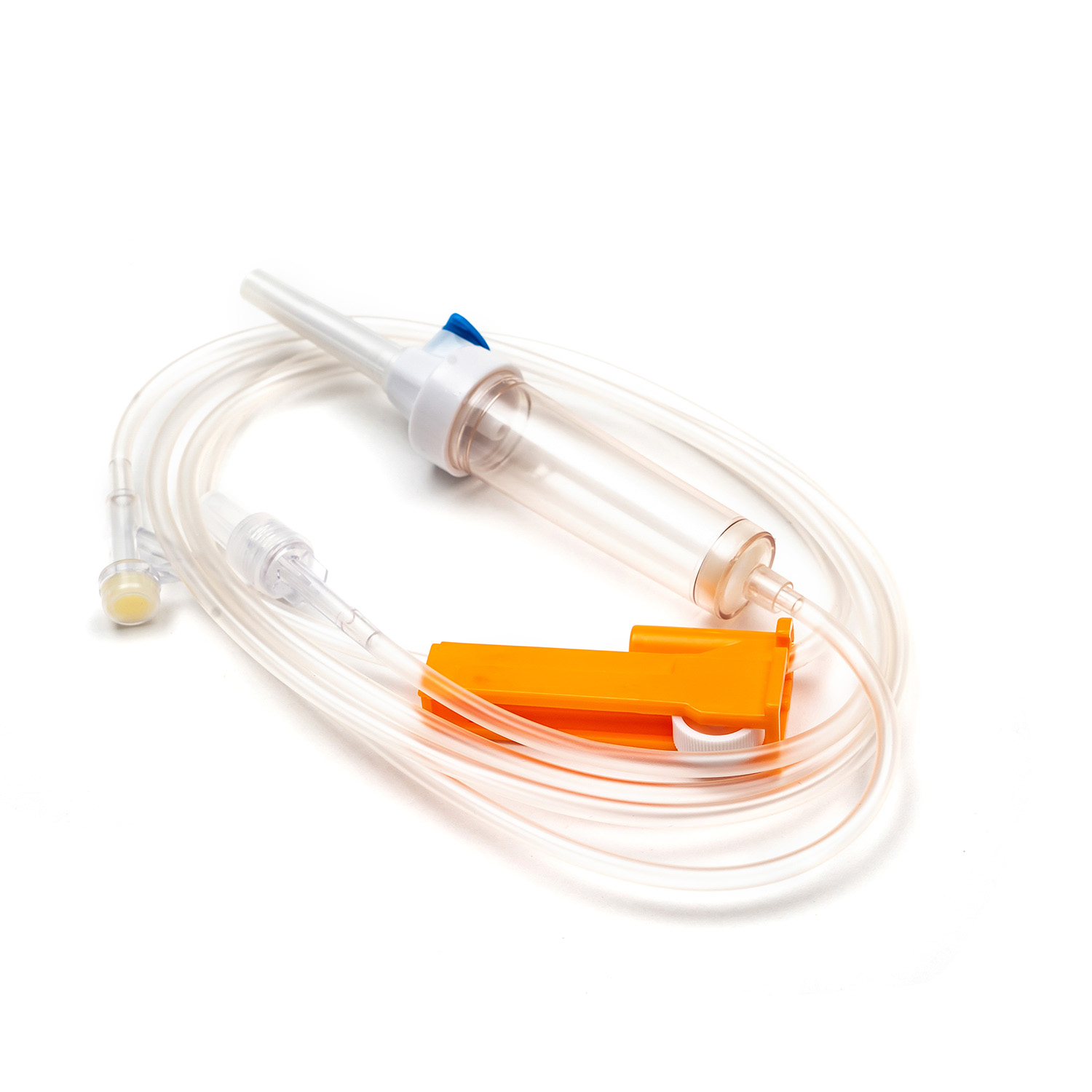
A complete disposable infusion set consists of multiple interconnected parts, each with a clear and independent function, working together to achieve stable and controllable liquid transmission. All components are made of medical-grade polymer materials, which are non-toxic, non-pyrogenic, and non-irritating to human tissues, ensuring biocompatibility during clinical use.
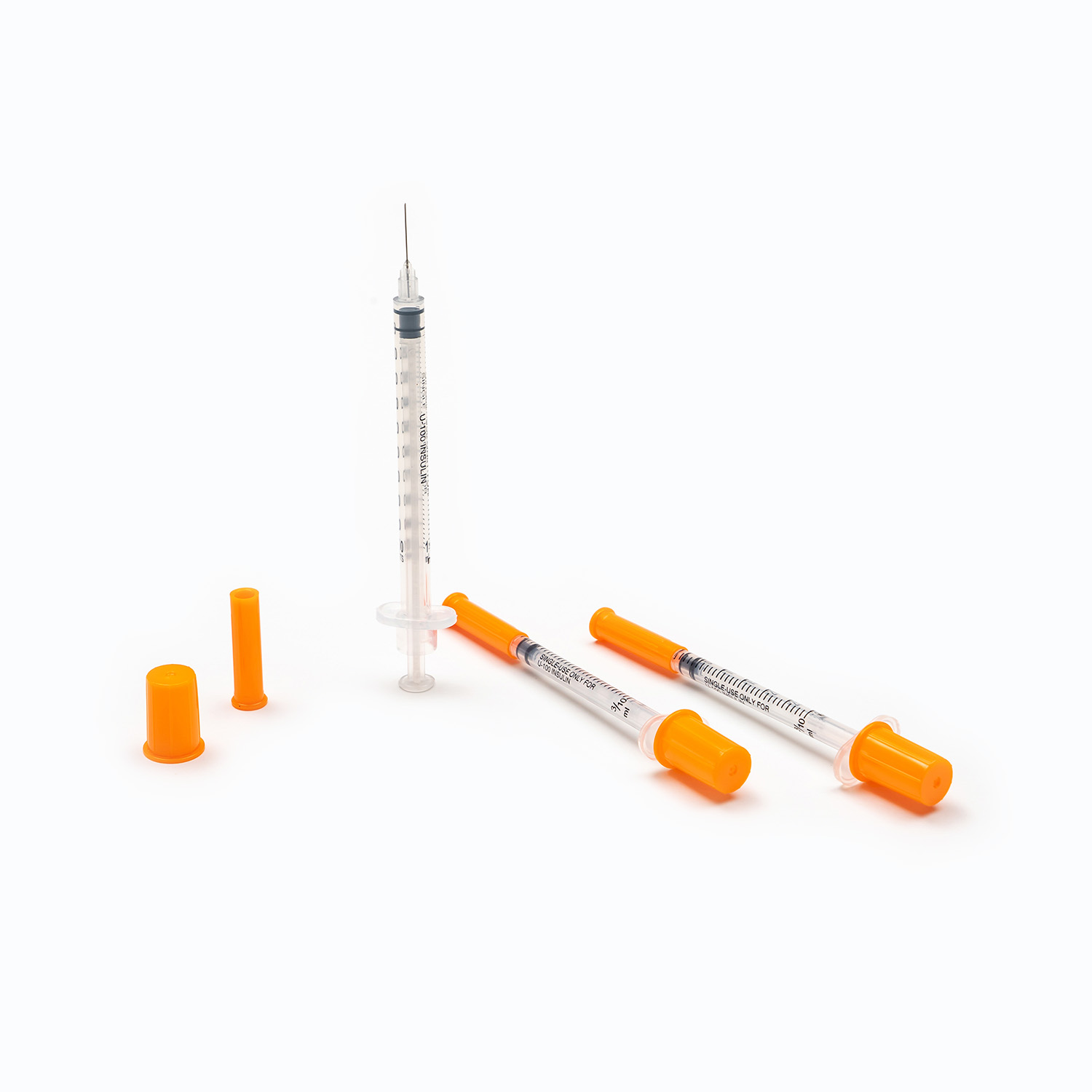
- The puncture needle assembly includes a stainless steel needle and protective sleeve, used to penetrate the patient’s vein and establish an infusion channel.
- The fluid inlet connector connects to the infusion bottle or bag, with an air filter or air vent to balance internal and external pressures and ensure smooth liquid flow.
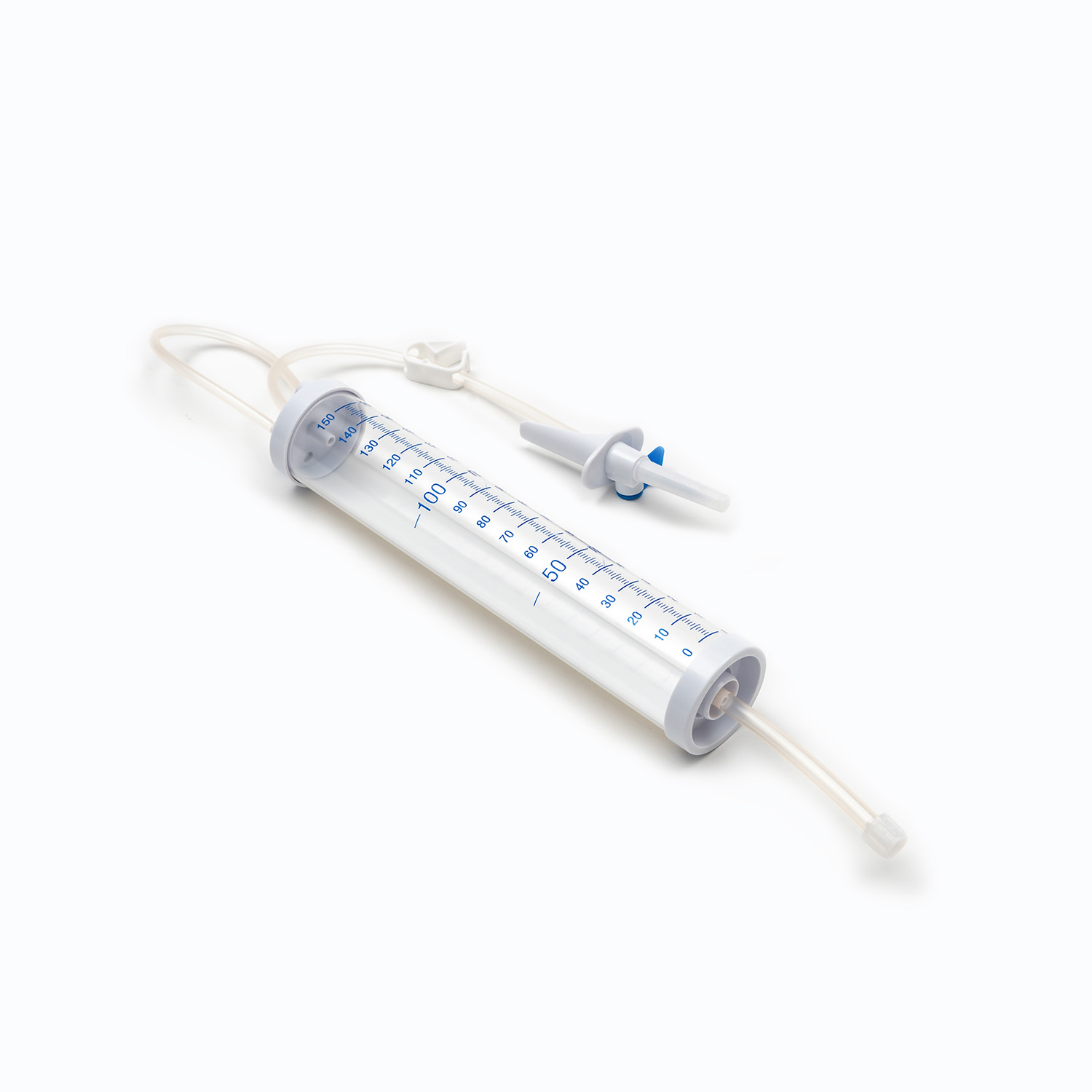
- The drip chamber is a transparent cavity that allows medical staff to visually observe and adjust the infusion rate by counting drops per minute.
- The infusion tube is a flexible pipeline that transports liquid, with sufficient length to meet clinical operation needs while preventing kinking and blockage.
- The flow regulator uses a roller or sliding mechanism to manually control the infusion rate, adapting to different treatment requirements and patient conditions.
- The liquid filter is a core component that removes particulate impurities, undissolved drug crystals, and microorganisms to reduce the risk of venous thrombosis and inflammation.
- The distal connector links to the indwelling needle or venous access, ensuring a sealed connection to avoid liquid leakage and external contamination.
Working Principles of Disposable Infusion Sets
Disposable infusion sets mainly rely on gravity pressure to achieve liquid transmission. The infusion container is hung at a certain height, and the liquid flows from high to low through the pipeline under the action of gravity. Medical staff adjust the flow regulator to control the cross-sectional area of the infusion tube, thereby changing the liquid flow rate and achieving precise infusion speed control.
In critical care and special treatment scenarios, some infusion sets are compatible with infusion pumps, which replace gravity with mechanical pressure to achieve more accurate and stable infusion, especially for high-risk drugs such as vasoactive agents, anesthetics, and chemotherapy drugs. The design of disposable infusion sets fully considers compatibility with infusion equipment, ensuring stable performance in both gravity and pump-assisted infusion modes.
Classification Based on Functional Design
To meet diverse clinical needs, disposable infusion sets are divided into multiple categories according to functional design, and medical staff select appropriate types based on treatment purposes, drug characteristics, and patient conditions.
- Basic gravity infusion sets: the most commonly used type, suitable for routine infusion of crystalloid fluids, antibiotics, and nutritional solutions.
- Filter-enhanced infusion sets: equipped with high-precision filters, used for chemotherapy drugs, blood products, and lipid emulsions to reduce particulate contamination.
- Light-proof infusion sets: made of light-shielding materials, used for drugs easily decomposed by light such as nitroglycerin and chemotherapeutic agents to maintain drug stability.
- Pediatric infusion sets: designed with smaller drip chambers and precise regulators, suitable for infants and young children to avoid excessive infusion speed and volume.
- Blood transfusion sets: with larger diameter pipelines and filters to prevent red blood cell damage and blood clot blockage.
Clinical Application Scenarios of Disposable Infusion Sets
Routine Inpatient Treatment
In general wards, disposable infusion sets are used for daily fluid replacement, anti-infective treatment, electrolyte balance adjustment, and symptomatic support. More than 80% of inpatients receive intravenous infusion therapy during hospitalization, making infusion sets one of the most frequently used medical devices in routine care. They are suitable for common diseases such as respiratory infections, digestive disorders, postoperative recovery, and chronic disease management, providing a stable and reliable way for drug delivery.
For long-term infusion patients, the single-use design avoids repeated disinfection and pipeline reuse, reducing the risk of superficial and deep venous infection. Medical staff replace the infusion set according to clinical guidelines to ensure the sterility and safety of the entire infusion process, which is an important measure for ward infection control.
Emergency and Critical Care
In emergency rooms and intensive care units, disposable infusion sets play a life-saving role. They are used for rapid fluid resuscitation in shock patients, continuous infusion of vasoactive drugs to maintain blood pressure, intravenous injection of rescue drugs such as epinephrine and atropine, and postoperative sedation and analgesia management. In emergency scenarios, rapid establishment of venous access and stable drug delivery directly affect the success rate of rescue, and the ready-to-use, sterile characteristics of disposable infusion sets greatly shorten the preparation time.
Critical patients often require multi-channel simultaneous infusion, and supporting disposable infusion sets can achieve independent control of each channel without mutual interference. High-precision filters effectively avoid micro-emboli caused by drug mixing and precipitation, reducing the risk of organ damage in critically ill patients with weak physical conditions.
Special Medical Fields
In oncology treatment, light-proof and filter-enhanced disposable infusion sets are specially used for chemotherapy drug infusion, protecting drug activity and preventing particulate contamination from stimulating blood vessels and causing phlebitis. In pediatric departments, special infusion sets with precise flow control reduce the risk of fluid overload in children, whose organ function is not fully developed, ensuring infusion safety.
In operating rooms, disposable infusion sets are used for intraoperative fluid replacement, anesthetic drug delivery, and blood transfusion support. Their sterile and sealed design meets the strict infection control requirements of operating rooms, and flexible pipelines adapt to various surgical positions without kinking or blockage. In outpatient transfusion rooms, they provide a safe and convenient way for short-term infusion treatment, supporting efficient operation of outpatient services.
Quality Standards and Safety Requirements for Disposable Infusion Sets
Sterility and Pyrogen-Free Standards
As a medical device in direct contact with the venous system, disposable infusion sets must meet strict sterility standards and be completely free of pyrogens, bacteria, fungi, and other pathogenic microorganisms. All products are sterilized by ethylene oxide or irradiation before leaving the factory, and the sterilization process is verified to ensure no residual toxins and no damage to device performance.
Pyrogen-free is a core safety indicator, as pyrogen entry into the blood vessel can cause fever, chills, and even life-threatening septic shock. Qualified infusion sets undergo strict pyrogen testing before packaging, and independent sterile packaging ensures no contamination during transportation and storage, maintaining sterility until the moment of opening for use.
Material Biocompatibility and Safety
Materials used in disposable infusion sets must pass systematic biological evaluation, including acute systemic toxicity tests, skin irritation tests, sensitization tests, and hemocompatibility tests. The materials shall not cause toxic reactions, tissue irritation, allergic reactions, or red blood cell damage in the human body, ensuring long-term contact safety.
Medical-grade polymer materials have good flexibility and tensile strength, not easy to break or fall apart during use. The inner wall of the pipeline is smooth to avoid liquid residue and bacterial adhesion, and is compatible with most clinical drugs without adsorption or chemical reaction that affects drug efficacy.
Performance Indicators and Clinical Adaptability
Qualified disposable infusion sets have stable and reliable performance indicators. The drip chamber has clear scales for accurate observation of infusion speed; the flow regulator has smooth adjustment and stable speed control without sudden changes; the filter has high filtration efficiency, effectively intercepting micro-particles while ensuring normal liquid flow; the pipeline has strong pressure resistance and no leakage or rupture under normal clinical use pressure.
| Performance Item | Safety Requirement | Clinical Significance |
|---|---|---|
| Sterility | Sterile, pyrogen-free | Prevent infection and fever reactions |
| Flow Regulation | Stable and adjustable | Ensure accurate drug delivery |
| Filtration Efficiency | Effectively intercept micro-particles | Reduce vascular irritation and emboli risk |
| Pipeline Performance | Leak-proof, kink-resistant | Guarantee continuous and stable infusion |
Standardized Operation and Usage Specifications of Disposable Infusion Sets
Pre-Use Inspection and Preparation
Before use, medical staff must conduct a comprehensive inspection of the disposable infusion set. First, check the outer packaging for damage, moisture, or contamination, and confirm the product is within the validity period. Damaged or expired products are strictly prohibited to avoid sterility failure and safety risks.
After confirming the packaging is intact, open it in a clean environment to avoid contamination. Connect the infusion set to the infusion container correctly, close the flow regulator, and squeeze the drip chamber to allow an appropriate amount of liquid to enter, exhausting all air from the pipeline. Air embolism is a serious infusion adverse event, and complete air exhaustion is a key step to ensure patient safety.
Intra-Use Operation and Monitoring
After successful venipuncture, fix the needle and adjust the flow regulator to set the infusion speed according to the doctor's advice, patient age, physical condition, and drug characteristics. Infants, elderly patients, and those with heart or kidney dysfunction require slower infusion speeds, while emergency fluid resuscitation needs rapid infusion as directed.
During infusion, medical staff regularly monitor the patient’s condition, infusion site, and pipeline status. Observe for local redness, swelling, pain, or liquid leakage, which may indicate needle displacement or phlebitis, requiring immediate adjustment or re-puncture. Monitor the infusion speed to prevent accidental speed changes caused by patient movement or pipeline compression, and ensure the drip chamber maintains normal liquid level to avoid air entering the blood vessel.
Post-Use Disposal and Management
Disposable infusion sets are for single use only and strictly prohibited from repeated use, cleaning, or disinfection for reuse. Reuse will damage the sterile state, reduce material performance, and greatly increase the risk of cross-infection, vascular damage, and drug contamination.
After use, the infusion set is classified as medical waste and disposed of following local medical waste management regulations. It is placed in a special sealed container to avoid needle stick injuries to medical staff and environmental pollution. Standardized disposal protects both medical staff and public health, forming a complete safe use chain of disposable infusion sets.
Common Problems and Preventive Measures in Using Disposable Infusion Sets
Infusion Speed Abnormality
Infusion speed too fast or too slow is a common problem, mainly caused by pipeline kinking, filter blockage, insufficient hanging height, or regulator failure. Preventive measures include checking pipeline smoothness before use, avoiding compression during placement, ensuring sufficient hanging height, regularly checking the drip chamber during infusion, and adjusting or replacing the infusion set promptly if abnormalities occur.
Venous Induced Adverse Reactions
Phlebitis and extravasation are common venous adverse reactions, related to drug irritation, infusion speed, and indwelling time. Using filter-enhanced or blood vessel-friendly infusion sets, controlling infusion speed reasonably, and replacing the infusion site regularly can effectively reduce the occurrence of such reactions. Once local redness, swelling, or pain occurs, stop infusion immediately and take targeted treatment measures.
Infection and Contamination Risks
The main infection risks come from damaged packaging, expired products, non-sterile operation, and reuse. Strictly implementing pre-use inspection, operating in a sterile environment, adhering to single-use principles, and standardizing waste disposal can reduce infection risk by more than 90%. Medical staff should receive regular training to master standardized operating procedures and ensure each step meets safety requirements.
Development Trends and Technological Innovations of Disposable Infusion Sets
With the development of medical technology, disposable infusion sets are continuously optimized and upgraded in terms of safety, functionality, and comfort. New materials with better biocompatibility and higher pressure resistance are gradually applied, reducing vascular irritation and improving pipeline durability. Intelligent infusion sets with integrated flow monitoring and alarm functions are used in critical care areas, automatically monitoring infusion speed and remaining liquid volume, and sending alerts for abnormalities to reduce manual monitoring pressure and human error.
Environmentally friendly disposable infusion sets made of biodegradable polymer materials are under development, reducing environmental pollution caused by medical waste while meeting clinical safety needs. Specialized infusion sets for targeted therapy, biological agents, and cell therapy drugs are also being developed, with higher precision filters and better drug compatibility to adapt to the development of precision medicine.
Global medical device standards are becoming more unified, promoting the quality consistency of disposable infusion sets worldwide and facilitating their application in medical systems of different countries and regions. The core development direction always focuses on improving clinical safety, reducing adverse events, and enhancing the ease of use for medical staff and comfort for patients.
Summary of Clinical Value and Safe Use Principles
Disposable infusion sets are fundamental and critical medical devices in modern healthcare, widely used in routine treatment, emergency rescue, critical care, and specialized therapy, providing a safe, effective, and convenient way for intravenous drug delivery. Their core value is reflected in preventing cross-infection, ensuring drug delivery accuracy, and improving the quality and safety of clinical care.
To maximize their efficacy, medical institutions and staff must adhere to three core principles: first, strictly select qualified products that meet national and international standards, and prohibit the use of unqualified, expired, or damaged devices; second, implement standardized operation procedures, including pre-use inspection, air exhaustion, speed adjustment, and real-time monitoring; third, strictly follow the single-use rule and standardize post-use disposal to avoid reuse and environmental pollution.
With technological progress and improved clinical standards, disposable infusion sets will continue to optimize and upgrade, bringing safer and more efficient infusion therapy experience to patients. As a cornerstone of intravenous therapy, their importance will continue to grow with the development of the medical industry, making irreplaceable contributions to global healthcare.



 English
English Français
Français русский
русский Español
Español