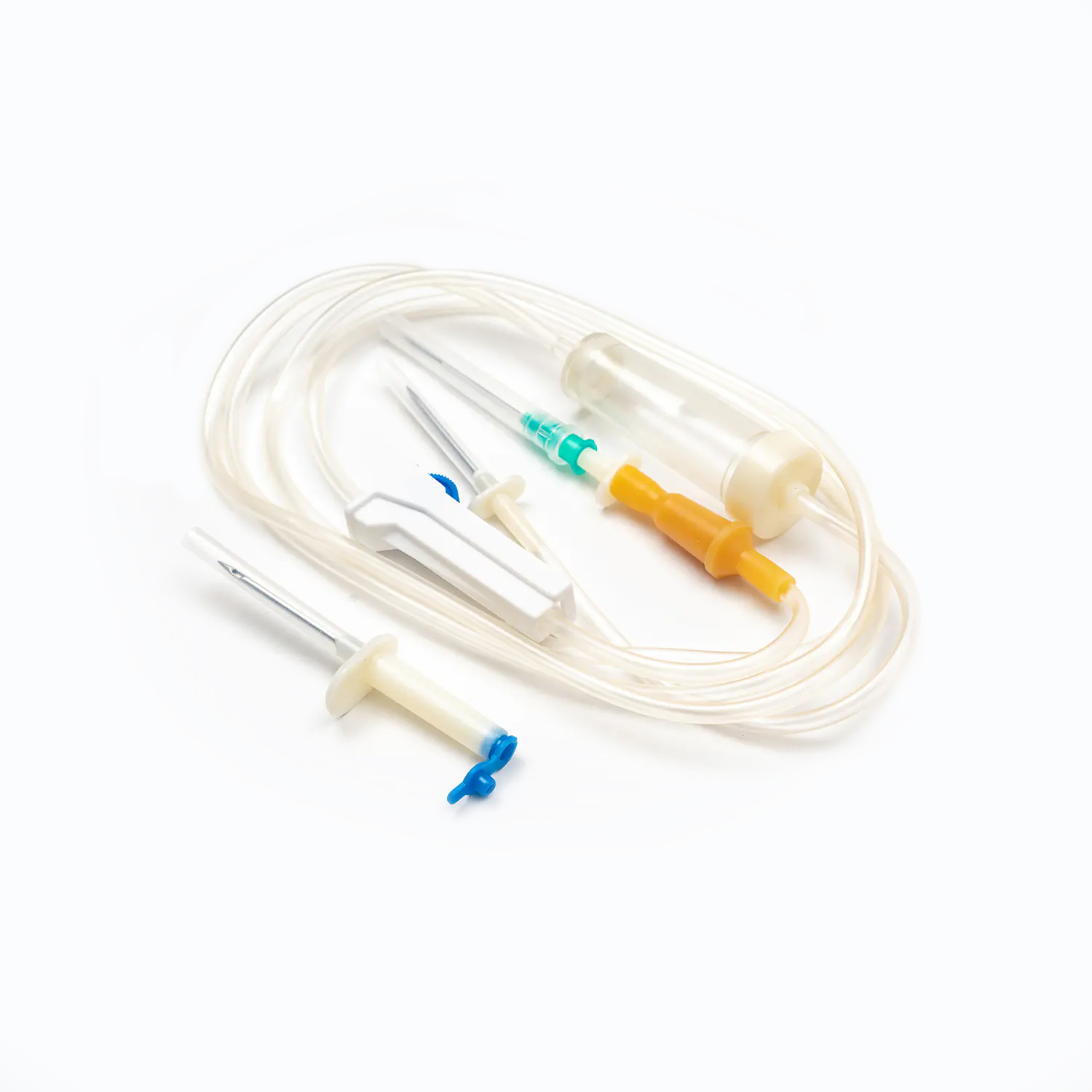
Top 5 Things to Check When Sourcing Disposable Syringes from China
Mar 08,2026
Disposable syringes are a crucial component of medical consumables, and their quality directly impacts medical safety and efficacy. Understanding the key specifications and quality standards of the product is essential when sourcing disposable syringes from China. This article provides professional guidance for medical procurement personnel from five core perspectives: product characteristics, certification standards, materials and manufacturing processes, packaging and transportation, and supply chain management.
Product Materials and Safety
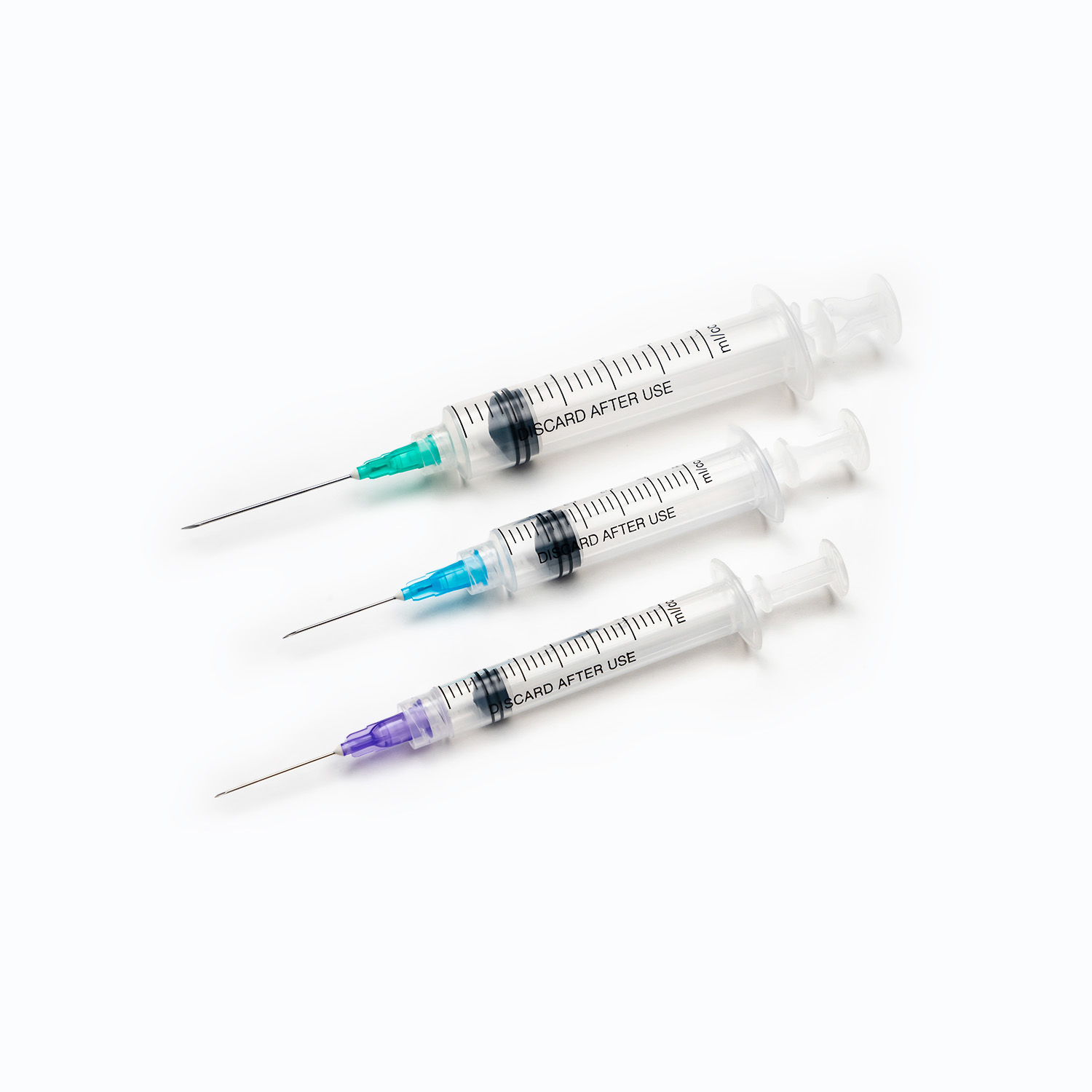
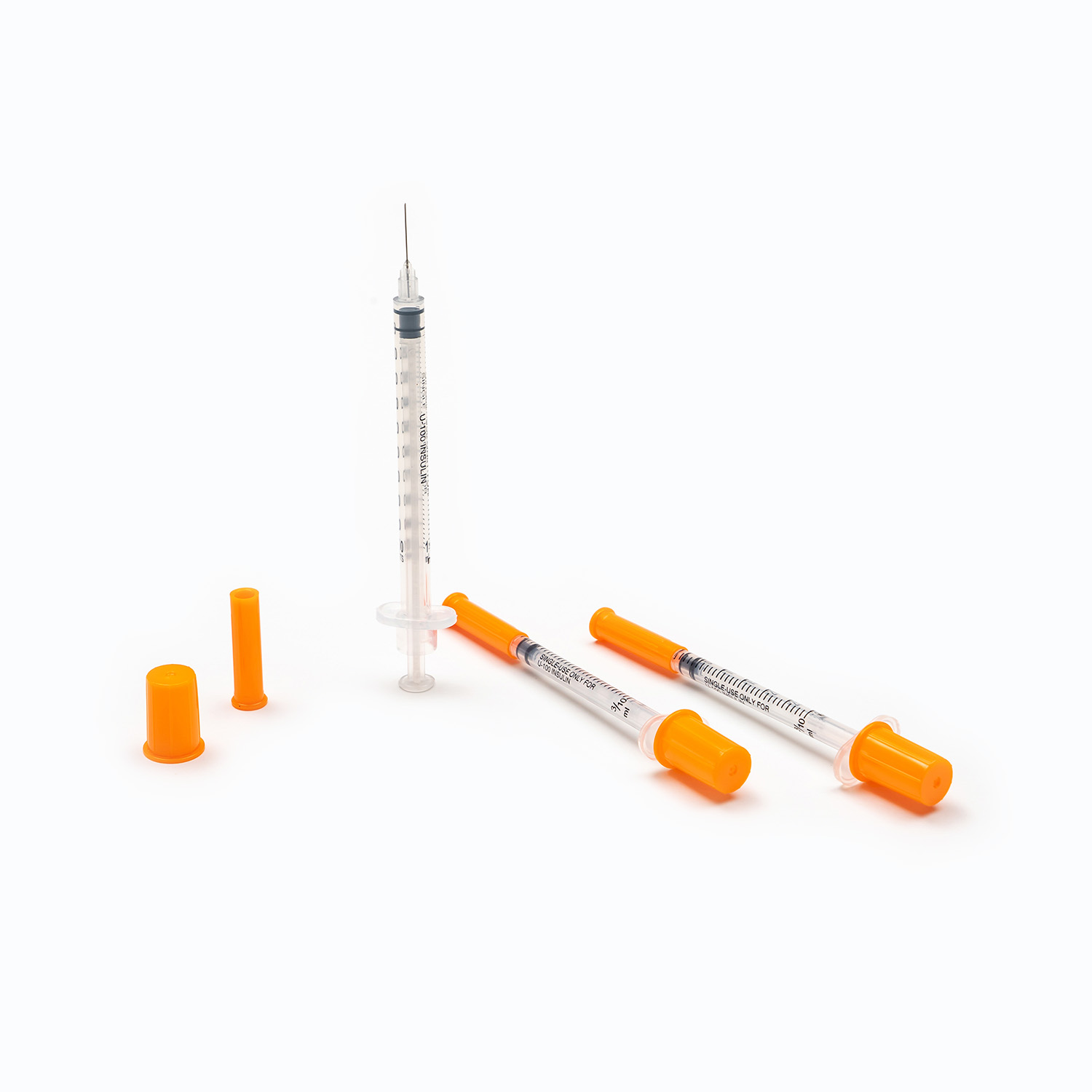
The core of disposable syringes lies in the safety and suitability of their materials. High-quality disposable syringes typically use medical-grade plastics or glass. These materials need to possess good biocompatibility, non-toxicity, and high-temperature resistance to ensure that no harmful substances are produced or deformation occurs during sterilization. Simultaneously, the metal material and plating process of the needle directly affect the smoothness of injection and the level of pain during puncture. During the procurement process, it is crucial to focus on the source of raw materials and the material testing reports provided by the supplier to ensure that the product will not pose a risk to patient health during use.
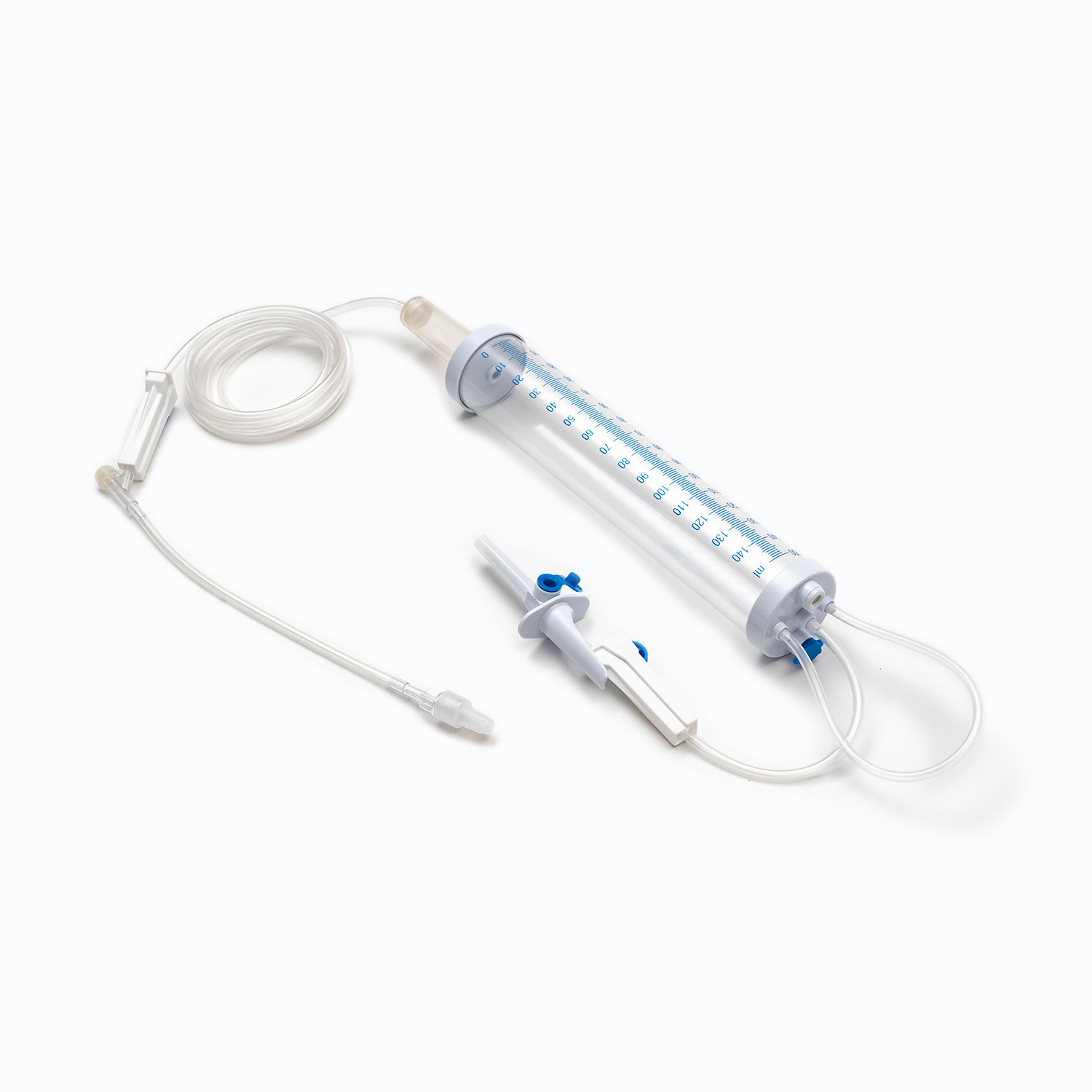
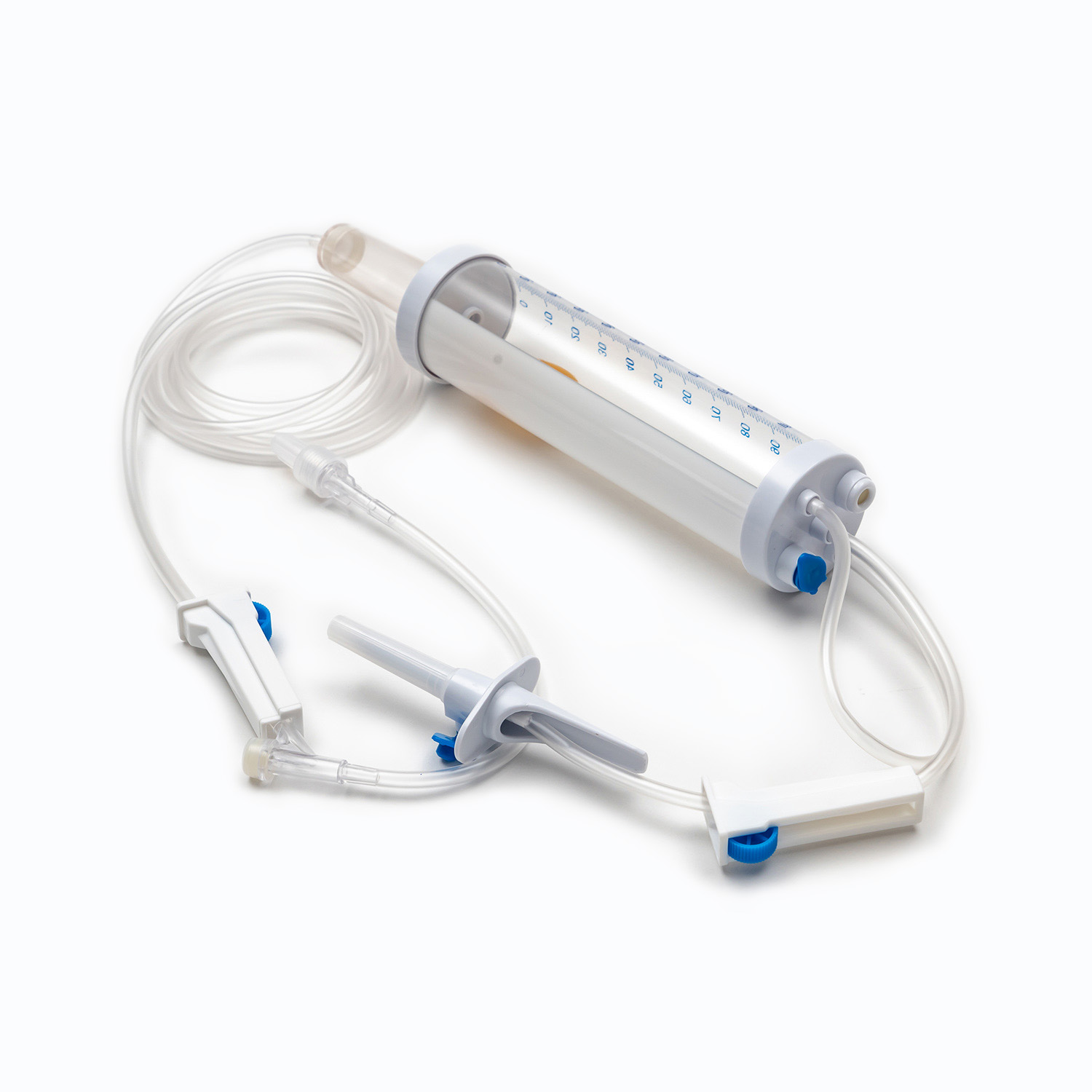
Design Precision and Functional Reliability
The design of disposable syringes not only affects the user experience but also the accuracy of drug delivery. The precision of key components such as the piston, syringe, and graduations must be strictly controlled to ensure the accuracy of each injection dose.
- Graduations should be clearly readable and abrasion-resistant.
- The seal between the piston and syringe should be excellent to prevent leakage or air bubbles.
- Safety lock design, needle protector, and anti-backflow function are essential additional features of modern disposable syringes.
- These features directly affect the safety of medical procedures and patient comfort.
Quality Certification and Regulatory Compliance
When procuring disposable syringes, it is crucial to prioritize product quality certification and regulatory compliance. Internationally recognized certification systems, such as ISO 13485 Medical Device Quality Management System, CE marking, and FDA registration, effectively demonstrate the reliability of supplier products. For domestic procurement, it is also necessary to pay attention to Chinese medical device registration certificates and product filing information.
- These certifications indicate that the product meets safety standards.
- They reduce legal and compliance risks during cross-border procurement.
- Whether a supplier possesses complete production qualifications and traceable production records is important for assessing product quality stability.
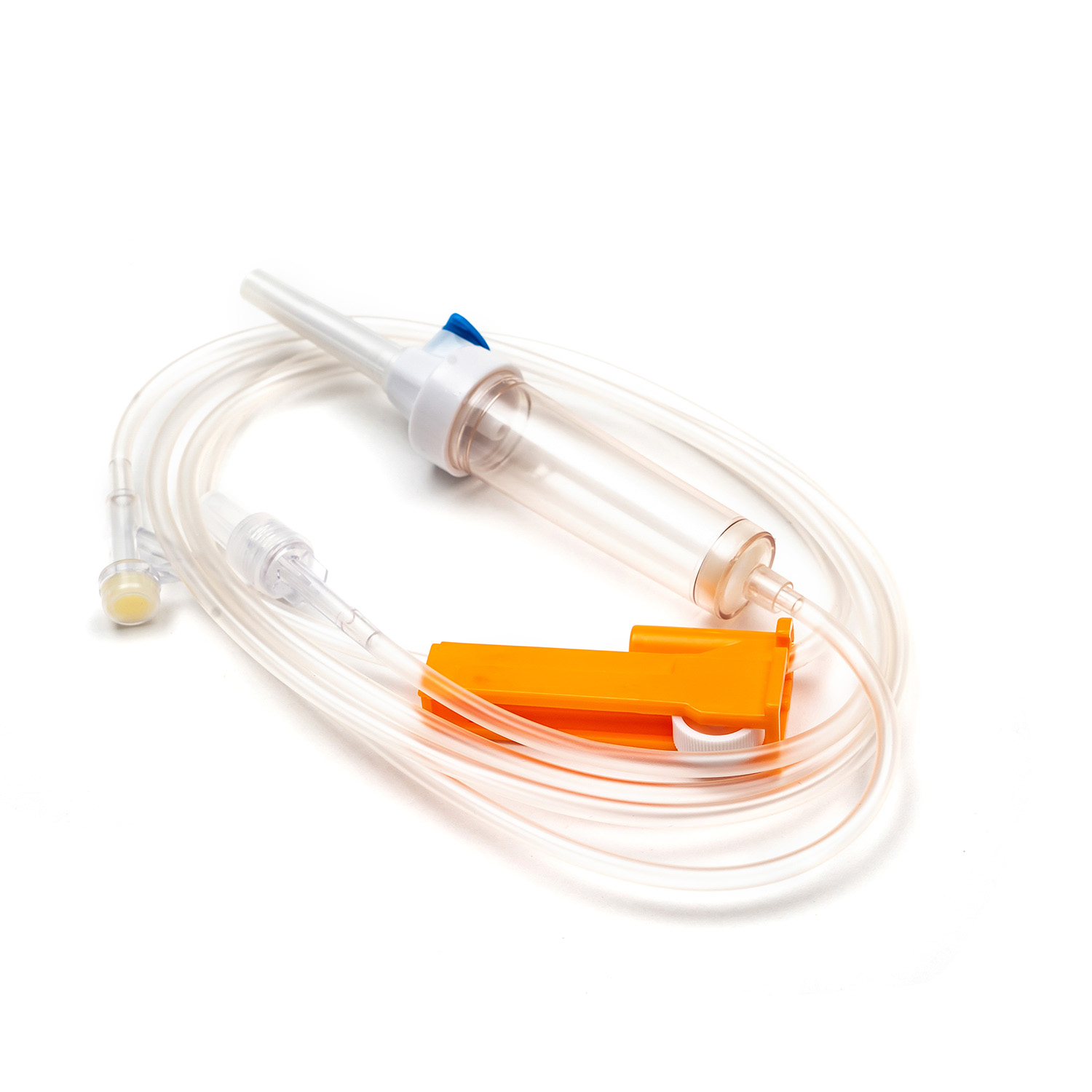
Packaging Design and Transportation Safety
Disposable syringes are high-risk medical consumables, and their packaging design directly affects product safety during transportation, storage, and use. Key considerations include:
- Sealed packaging and moisture-proof design.
- Aseptic processing to maintain sterility.
- Breakage-resistant and pressure-resistant packaging materials.
- Packaging should facilitate warehousing management and efficient use while minimizing waste and operational risks.
Supplier Capabilities and After-Sales Support
In addition to product quality, supply chain capabilities and after-sales support are crucial when selecting suppliers:
- Advanced production equipment and stable manufacturing processes affect product consistency.
- Batch management capabilities ensure reliable output quality.
- Suppliers should provide complete testing reports and quality tracking systems.
- Effective after-sales service, including technical support or product replacement, reduces operational risks for medical institutions.
FAQ
Q: What are the requirements for sterile packaging of disposable syringes?
A: Sterile packaging should ensure the product remains sterile throughout transportation and storage. Packaging materials must be moisture-proof, breakage-proof, and able to withstand high-temperature sterilization.
Q: How to confirm syringe accuracy during procurement?
A: Review the supplier's factory inspection report and production process, focusing on the accuracy of the scale, piston sealing, and medication backflow.
Q: How does the material selection of disposable syringes affect their use?
A: The material determines high-temperature resistance, chemical resistance, and biocompatibility, directly impacting injection smoothness, patient comfort, and long-term storage safety.
Q: What qualifications should be considered when selecting a supplier?
A: Focus on medical device registration certificates, ISO 13485 quality system certification, CE or FDA certification, as well as production traceability records and quality control capabilities.
Q: How can we ensure that disposable syringes are not contaminated during transportation?
A: They need to be sealed and individually packaged, and the transportation environment must be dry and clean to prevent packaging damage or moisture that could affect sterility.



 English
English Français
Français русский
русский Español
Español