Luer Lock vs Luer Slip Syringes: Types, Uses, and What to Specify When Ordering
Mar 22,2026
Disposable syringes are among the highest-volume consumables in any hospital or clinic purchasing program. Their specification seems simple — volume, sterile, disposable — but the details of tip configuration, barrel material, graduation accuracy, and needle compatibility affect clinical workflow and patient safety in ways that aren't always obvious until a mismatch causes a problem. This guide covers the main syringe categories, the luer connection standard that determines needle compatibility, and the specification points that matter most for bulk procurement.
The Luer Connection Standard
Nearly all modern syringes and needles connect through the luer standard, an ISO-defined taper fit (ISO 594-1 and ISO 80369) that ensures compatibility across manufacturers. The luer taper is a 6% taper — a slight conical shape that creates a friction or mechanical lock when mated parts are pushed together.
Within the luer standard, two connection types determine whether a needle or device simply slides onto the syringe tip or locks mechanically:
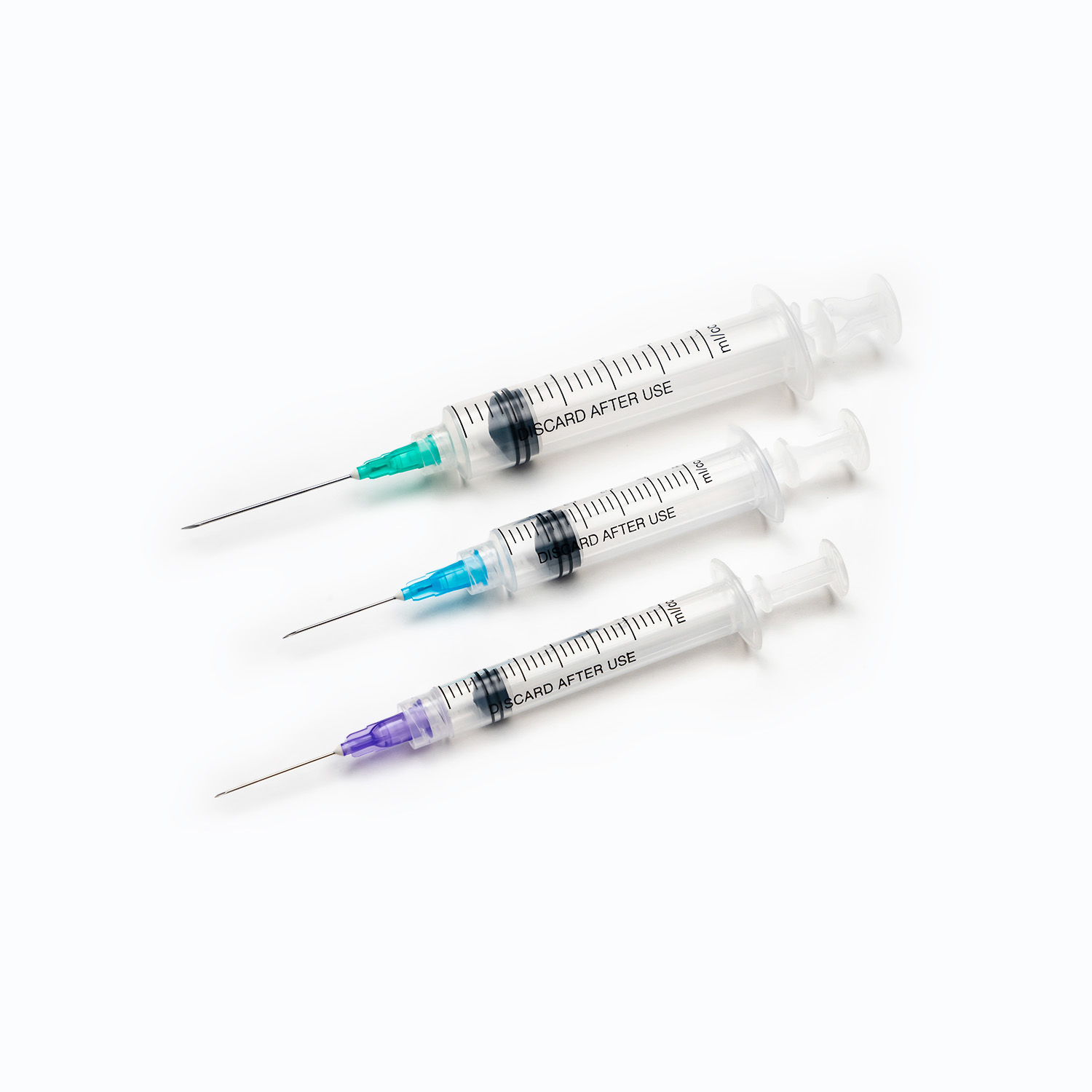
Luer Slip (Plain Tip)
A luer slip syringe has a smooth tapered tip. A needle hub slides over the tip and is held in place by the friction fit of the luer taper alone. This connection is fast to attach — the needle goes on with a push — and fast to detach. For high-speed clinical settings like vaccination programs, mass injection campaigns, or situations where needle changes happen frequently, the speed advantage of Luer slip is meaningful. The tradeoff is that the friction connection can separate under pressure, which is a problem for injections requiring significant force or for high-pressure infusion applications where the needle connection must hold against back-pressure.
Luer Lock
A luer lock syringe adds a threaded collar around the tip. The needle hub engages both the taper fit and the threaded collar, which is rotated to create a positive mechanical lock. The connection cannot be pulled apart without unscrewing the collar. Luer lock is mandatory for any application involving significant pressure or force: arterial lines, power injection (CT contrast), viscous medications that require pressure to inject, and any situation where accidental needle separation would cause injury or medication loss. In most hospital clinical settings, luer lock has become the default precisely because the security of the connection outweighs the minor additional step of screwing the collar.
The two types are physically compatible at the taper connection — a luer lock needle will fit on a luer slip syringe (though the collar can't engage), and a luer slip needle will fit on a luer lock syringe. But using a luer lock needle on a luer slip syringe without the collar engaged negates the lock's purpose, and using a luer slip needle on a luer lock syringe means the collar can't properly secure the connection. For consistent clinical practice and to avoid staff confusion, stocking Luer Lock syringes and Luer Lock needles as the standard across a facility simplifies the system.
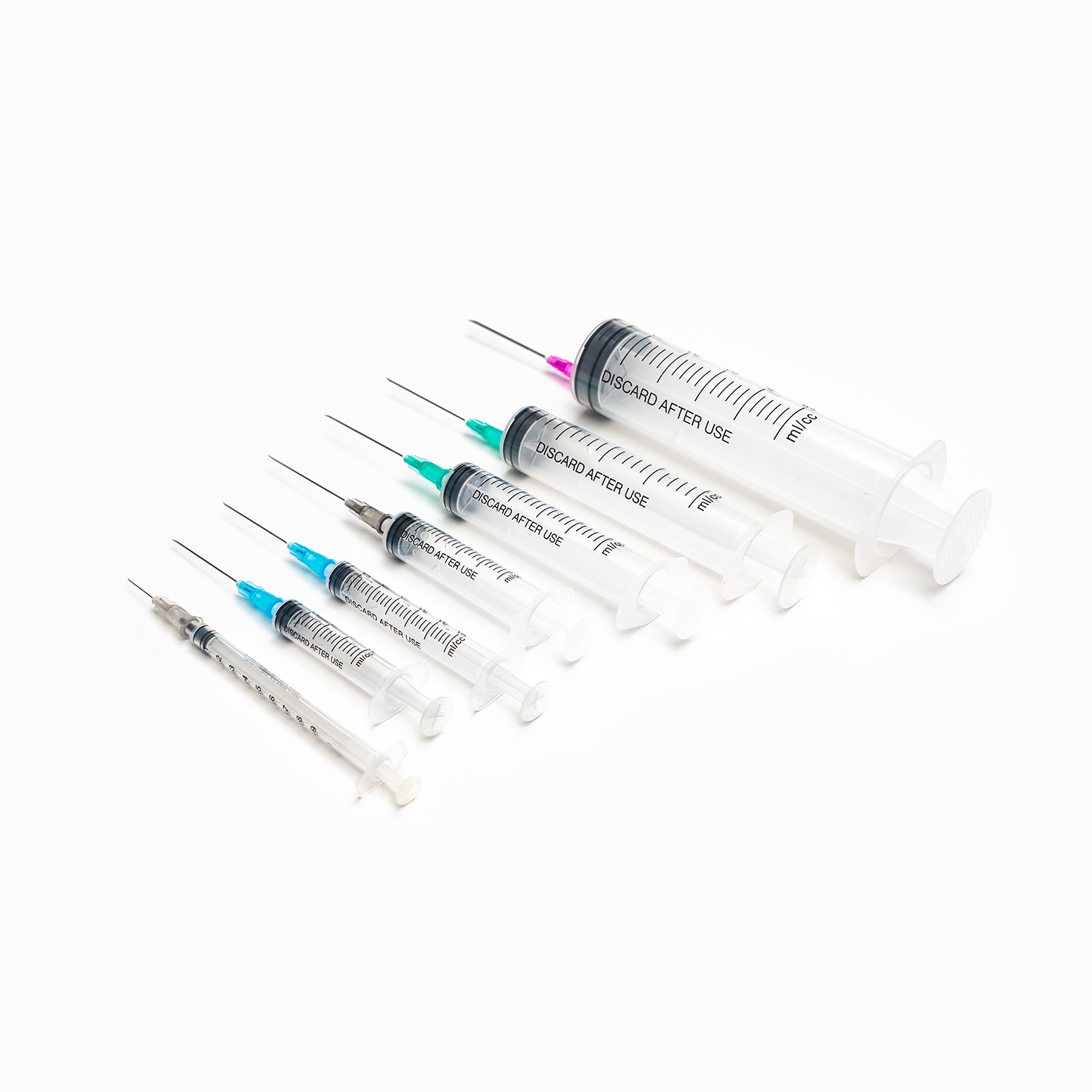
Syringe Volume Configurations and Their Clinical Uses
Syringe volume selection follows the general principle of choosing the smallest volume syringe that accommodates the required dose for accuracy. Larger syringes have coarser graduation markings — a 50mL syringe might have markings every 2mL, making a 1.5mL dose reading imprecise. A 3mL syringe for a 1.5mL dose allows reading to 0.1mL.
| Volume | Typical Graduation | Primary Clinical Uses | Notes |
|---|---|---|---|
| 1 mL | 0.01 mL | Intradermal injections, allergy testing, concentrated medications | Tuberculin syringe; also standard for many vaccine doses |
| 2 mL | 0.1 mL | Subcutaneous and IM injections, small-volume medications | Common in pediatric dosing and vaccination programs |
| 3 mL | 0.1 mL | Most common IM and SC injections, general medication doses | The most widely used general-purpose syringe volume |
| 5 mL | 0.2 mL | IV bolus doses, blood draws for small samples, and medication mixing | Good for doses in the 3–5 mL range |
| 10 mL | 0.5 mL | IV medication administration, flush procedures, and blood sampling | Standard for IV line flushing; heparinized flush protocols |
| 20 mL | 1 mL | Larger-volume IV medications, specimen collection, and irrigation | Used in procedures requiring moderate fluid volumes |
| 50 mL | 2 mL | Enteral nutrition, large-volume irrigation, and pump reservoir filling | Often used in syringe pumps for continuous infusion |
| 60 mL | 2 mL | Irrigation, enteral feeding, wound care | Tip often catheter-tip style for irrigation applications |
Eccentric vs Concentric Tips
Beyond the luer lock/slip distinction, syringe tip position affects clinical use in specific situations. Most syringes have a concentric tip — the tip is centered on the end of the barrel. Eccentric tip syringes have the tip positioned off-center, closer to the edge of the barrel. This allows the syringe barrel to lie nearly flat against the patient's skin surface during injection, which is the preferred technique for intradermal injections (where the needle must enter the skin at a very shallow angle, nearly parallel to the surface) and for venipuncture in some protocols. Eccentric tip syringes are a niche product, but should be in any procurement catalog that supports dermatology, allergy testing, or intravenous access programs.
Three-Part vs Two-Part Construction
Syringes are manufactured in two-part (barrel and plunger, no rubber stopper) and three-part (barrel, plunger rod, rubber plunger tip) configurations. Three-part syringes — the standard for most clinical applications — use a rubber tip on the plunger that creates a smooth, consistent seal against the barrel wall. This seal provides predictable plunger resistance, accurate dead-space control, and reliable aspiration. The rubber tip also allows the syringe to draw back smoothly for aspiration checks before injection.
Two-part syringes (all-plastic construction without rubber) are used in specific applications where rubber components are contraindicated, such as certain drug preparations where the plasticizers in rubber could interact with the medication. For general clinical use, three-part syringes are the standard specification. When sourcing syringes, confirm the plunger construction and rubber tip material — natural rubber versus synthetic rubber matters for drug compatibility in certain applications.
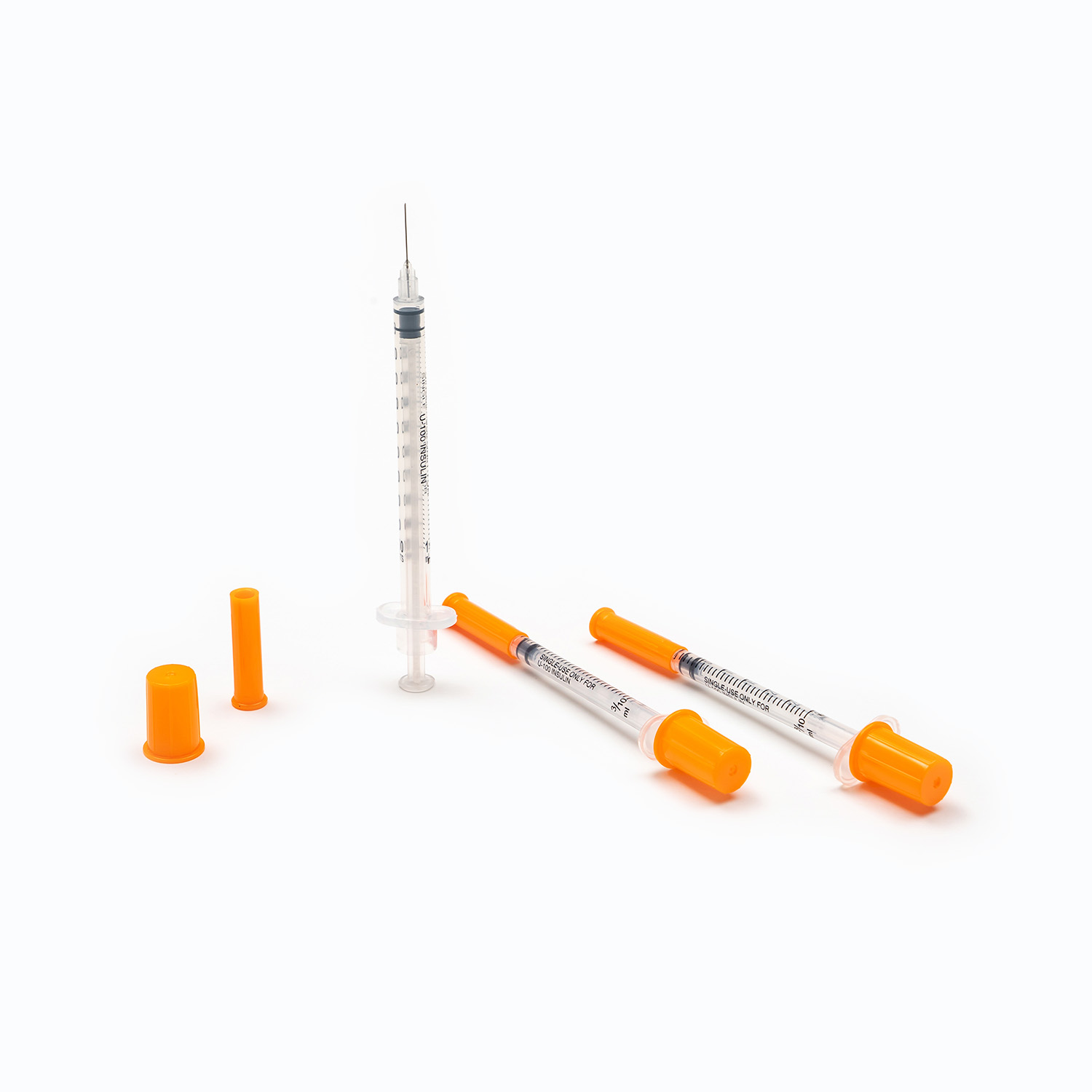
Insulin Syringes: A Distinct Category
Insulin syringes are not general-purpose syringes and should be specified and stocked separately. The key distinguishing features are the volume scale (U-100 markings in insulin units rather than mL) and the permanently attached needle (insulin syringes are supplied with a needle attached and are not compatible with separate needle attachment). The needle gauge for insulin syringes is also finer than general-purpose needles — typically 28–31G — to minimize injection discomfort for subcutaneous administration.
U-100 insulin syringes are calibrated for U-100 insulin (100 units per mL), which is the global standard concentration. Some markets also use U-40 insulin, which requires U-40 syringes — these are not interchangeable, and using a U-100 syringe to administer U-40 insulin (or vice versa) results in dosing errors. For facilities or procurement programs serving markets where U-40 insulin is in use, both syringe types must be stocked and clearly differentiated in storage and dispensing.
What to Specify and Verify When Ordering
A complete syringe specification for procurement purposes should include: volume, tip type (luer lock or luer slip), barrel material (polypropylene is standard), plunger construction (three-part with rubber tip), sterility (EO sterilization, ISO 11135), graduation accuracy (ISO 7886-1 defines ±4% for Class A, ±8% for Class B), needle included or syringe-only, packaging (individual peel-pack or bulk), and shelf life.
For bulk procurement from manufacturers rather than distributors, additional quality verification points include: ISO 13485 quality management system, CE marking or equivalent market authorization, batch traceability documentation, and sterilization validation records. Syringe-and-needle combination products have a higher complexity quality profile than syringe-only products because the needle gauge, bevel geometry, and coating (siliconized needles reduce insertion force) all affect clinical performance independently.
Lot-to-lot consistency is a practical concern that isn't always addressed in initial supplier qualification. Requesting test data from multiple production batches — rather than a single sample batch — gives a more representative picture of whether graduation accuracy, plunger force, and sterility assurance hold consistently across production. This is especially relevant for higher-volume consumables where any quality inconsistency affects large patient numbers before it's identified and corrected.
Frequently Asked Questions
Are Luer Lock and Luer Slip syringes interchangeable at a facility level?
Operationally, most facilities standardize on Luer Lock for IV and injection use because the secure connection is safer, and the minor speed disadvantage is not clinically significant for most applications. Running both types in a facility creates inventory management complexity and the risk of staff using the wrong type for a given application. The main scenario where luer slip remains preferred is mass vaccination or high-throughput injection programs where the speed of connection matters; for standard ward and clinical use, luer lock is the practical default that most contemporary procurement guidelines recommend.
What gauge needle is most commonly used for intramuscular injections?
Intramuscular injections typically use 21–23G needles, with the specific choice depending on medication viscosity and patient body habitus. Thicker medications (some vaccines, oil-based preparations) require a larger-gauge needle (21G) to flow adequately; thinner medications can use a finer gauge (23G) for less patient discomfort. Needle length for IM injections varies with injection site and patient adipose tissue depth — 25mm (1 inch) is standard for the deltoid in adults of normal weight; 38mm (1.5 inch) for the vastus lateralis or gluteus medius. Needle length selection is a clinical decision that should be documented in the facility's injection technique protocols.
How should syringes be stored, and what affects shelf life?
Sterile single-use syringes are stable for 3–5 years from the date of manufacture when stored under the recommended conditions: cool (below 30°C), dry (relative humidity below 75%), dark environment away from UV exposure, and away from chemical fumes that could degrade packaging integrity. High temperature and humidity are the primary factors that can degrade the packaging seal or the rubber plunger tip before the nominal expiry date. First-in-first-out rotation of syringe stock is essential in high-volume environments to prevent expired waste. Any package with compromised seal integrity — tears, punctures, or moisture evidence — should be discarded regardless of the printed expiry date.
Syringes | Insulin Syringes | Infusion Sets | Blood Lancet Needle | Contact Us



 English
English Français
Français русский
русский Español
Español